Did all of the particles go straight through the gold foil?
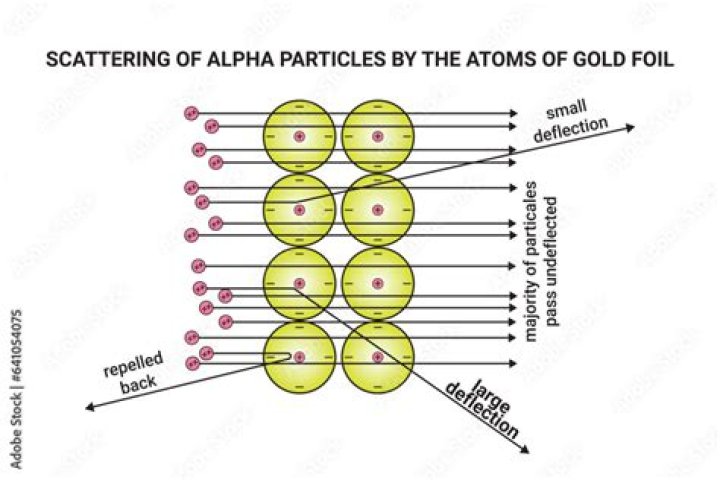
Most of the alpha particles did pass straight through the foil. The atom being mostly empty space. A small number of alpha particles were deflected by large angles (> 4°) as they passed through the foil. Alpha particles were known to be much, much more dense than gold.
Why did some particles go through the gold foil?
In Rutherford’s gold foil experiment, the particles of which most passed straight through the gold foil were alpha particles, which are positively charged. The few alpha particles that were deflected back or to one side were being repelled by nuclei, which are also positively charged.
Which particles are passed through the gold foil?
Most alpha particles passed straight through the gold foil, which implied that atoms are mostly composed of open space. Some alpha particles were deflected slightly, suggesting interactions with other positively charged particles within the atom.
What particles did Rutherford Fire at gold foil?
Rutherford’s gold foil experiments (and other metal foil experiments) involved firing positively charged alpha particles at a piece of gold/metal foil. The alpha particles that were fired at the gold foil were positively charged.
Why do most alpha particles fired through a piece of gold foil emerge almost Undeflected?
Why do most alpha particles fired through a piece of gold foil emerge almost undeflected? The massive alpha particles blast through the majority of the space in the gold that is occupied by low mass electrons. The mass of the atom is concentrated in a positively charged core.
Why did Rutherford use gold foil instead of any other metal?
For the scattering experiment, Rutherford wanted a metal sheet which could be as thin as possible. Gold is the most malleable of all known metals. It can easily be converted into very thin sheets. Hence, Rutherford selected a gold foil for his alpha-ray scattering experiment.
Why do most of the α particles in Rutherford’s experiment go through gold foil without being deflected?
This is due to the fact that like charges repel each other. As the positively charged alpha particle would fly through the foil it would come in proximity with the positively charge nucleus of the atom. This in turn either deflected the particle or adjusted its path.
Why do most of the particles passed through the gold foil without getting deflected?
Most of the space inside the atom is empty because most of the α-particles passed through the gold foil without getting deflected. Very few particles were deflected from their path, indicating that the positive charge of the atom occupies very little space.
Why did most of the particles pass straight through the gold foil?
Educators go through a rigorous application process, and every answer they submit is reviewed by our in-house editorial team. In Rutherford’s gold foil experiment, the particles of which most passed straight through the gold foil were alpha particles, which are positively charged.
Why did Rutherford put alpha particles in gold foil?
It’s not necessarily straightforward, at least to me, why you would think these alpha particles would just go straight through the gold foil. So what Rutherford, at the time, was doing was, he was testing the plum pudding model.
What happens when you hit gold foil with a bullet?
But it turned out that for every one in… one in 20,000 alpha particles, or some crazy-tiny number like that, for every one in 20,000 alpha particles, he saw the particles hit the gold foil and bounce back. And that’s crazy, right? That’s exactly what you don’t expect when you hit a piece of tissue paper with a bullet.
Why do most alpha particles pass straight through the gold?