Does copying of copyrighted material comes under plagiarism?
It is illegal to copy large sections of someone else’s copyrighted work without permission, even if you give the original author credit. Just make sure to give the author credit so you won’t be guilty of plagiarism! The courts assign consequences for copyright infringement.
What is considered plagiarism in a research paper?
Plagiarism is presenting someone else’s work or ideas as your own, with or without their consent, by incorporating it into your work without full acknowledgement. All published and unpublished material, whether in manuscript, printed or electronic form, is covered under this definition.
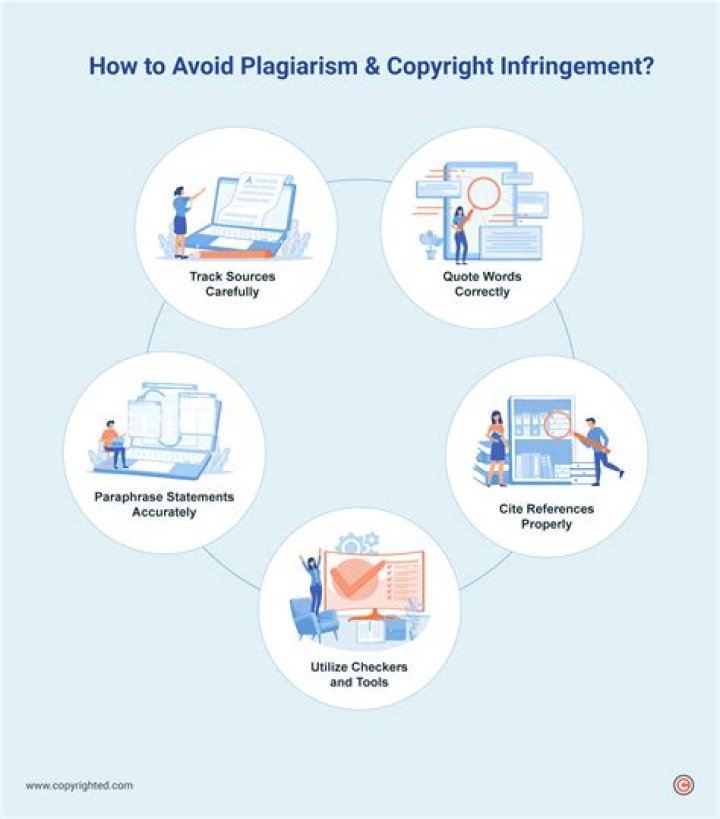
How do I copy and paste without plagiarizing?
Avoid Copy and Paste Plagiarism
- You can avoid Copy and Paste plagiarism by using quotes.
- There are two ways to quote: You can put quotation marks around a sentence in the text. OR. If your quote takes up more than four typed lines, you can use block quotations. Block quotations are indented from the main body of the page.
What is the difference between plagiarism and copying?
As nouns the difference between copying and plagiarism is that copying is (countable) an instance of the making of a copy while plagiarism is (uncountable) the act of plagiarizing: the copying of another person’s ideas, text or other creative work, and presenting it as one’s own, especially without permission.
How many words can you copy without plagiarizing?
Plagiarism: taking any sequence of more than three words without citing is stealing work from others. Taking an idea, image (photograph, table, or graph) without citing is also plagiarism and may also violate copyright laws.
Why are warning labels included in food packages?
Warning labels should be including in food packages because they will help people take control of what they consume and food industries will have to provide healthier options. People will become more aware of what they are eating if there are labels on food packages.
Is it difficult to comply with FDA labeling requirements?
Indeed, ensuring that all information concerning that particular dietary supplement is in compliance with FDA labeling requirements, may be difficult and often very onerous. As a matter of fact, labeling errors may result in FDA enforcement action which could tarnish your company’s reputation and business.
What are the labeling requirements for GMP devices?
The QA program must be adequate to ensure that labeling meets the GMP device master record requirements with respect to legibility, adhesion, etc., and ensure that labeling operations are controlled so that correct labeling is always issued and used. Labeling includes equipment labels, control labels, package labels, directions for use.
What are the labeling requirements for a product?
Labeling specifications are: engineering drawing and/or artwork for each label, appropriate inspection or control procedures, and appropriate procedures for attaching the labels. All procedures, drawings, and artwork must have the name of the preparer, an approval signature, and a date.