Does gold react with oxygen water or acid?
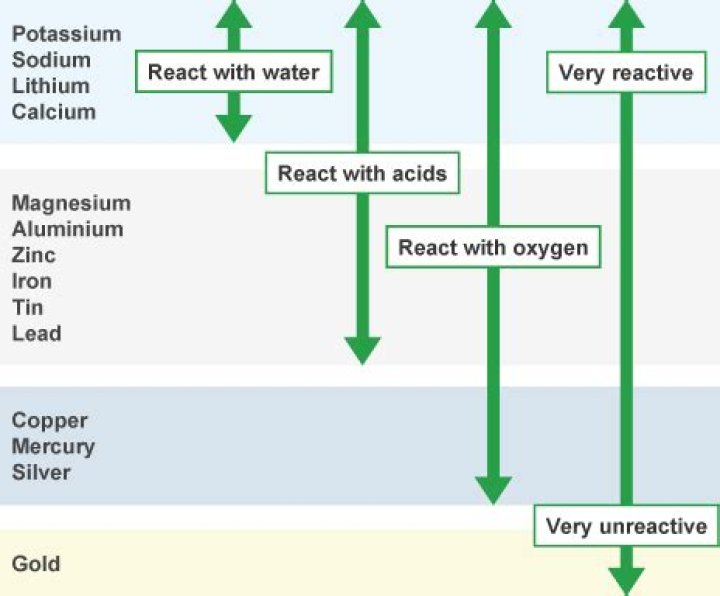
Gold is one of the least reactive elements on the Periodic Table. It doesn’t react with oxygen, so it never rusts or corrodes. Gold is unaffected by air, water, alkalis and all acids except aqua regia (a mixture of hydrochloric acid and nitric acid) which can dissolve gold.
Is gold and silver react with oxygen to form basic oxide?
Metals such as potassium and sodium react so vigorously that they catch fire if kept in the open. Copper does not burn, but the hot metal is coated with a black coloured layer of copper(II) oxide. Silver and gold do not react with oxygen even at high temperatures.
What type of oxide is formed when a metal react with oxygen?
When a metal reacts with oxygen, a metal oxide forms. The general equation for this reaction is: metal + oxygen → metal oxide. Rust is a form of iron oxide and it forms slowly when iron is exposed to air.
Is an oxide a base or acid?
An oxide that combines with water to give an acid is termed as an acidic oxide. The oxide that gives a base in water is known as a basic oxide. An amphoteric solution is a substance that can chemically react as either acid or base….Trends in Acid-Base Behavior.
| 1 | Li |
|---|---|
| 2 | Be |
| 3 | B |
| 14 | C |
| 15 | N |
Does oxygen react to form basic oxide?
Metals react with oxygen to form basic oxides. Metallic oxides are basic in nature because they react with dilute acids to form salt and water. They also react with water to form metal hydroxides which are alkaline in nature because these metal hydroxides release OH− ions in solution.
What happens when a metal reacts with oxygen?
Many metals react with oxygen to form metal oxides. Potassium and sodium are soft metal which are easily cut exposing a shiny surface which changes to dull rapidly. The change from shiny to dull is called tarnishing.
Which metal reacts vigorously with oxygen and water?
Sodium is the metal that reacts with oxygen and water vigorously.
What kind of reaction does gold have with oxygen?
Gold does not react directly with oxygen, however if you succeed in getting gold oxide indirectly, it decomposes at 205C. The oxide is neither acid nor base however gold ions are acidic (like protons are acidic) What properties is possessed by gold but not possessed by silver?
What is the formula for gold ( III ) oxide?
From Wikipedia, the free encyclopedia Gold (III) oxide (Au 2 O 3) is an inorganic compound of gold and oxygen with the formula Au 2 O 3. It is a red-brown solid that decomposes at 160 °C. According to X-ray crystallography, Au 2 O 3 features square planar gold centers with both 2- and 3-coordinated oxides.
When do metals react with oxygen do they form basic compounds?
Basic oxide: Metals react with oxygen to give basic compounds of oxygen. These compounds are usually ionic in nature. Group 1, 2 and lanthanides form basic compounds of oxygen when they react with dioxygen. During the formation of these compounds, a large amount of energy is released. These compounds readily react with water except few exceptions.
How are oxygen oxides different from other oxides?
Oxygen is highly reactive in nature. They react with metals and non-metal to form oxides. What are Oxides? The classification of oxides is done into neutral, amphoteric and basic or acidic based on their acid-base characteristics. An acidic oxide is an oxide which when combined with water gives off an acid.