Does silver nitrate dissociate in water?
C. In the dissolution process we break strong ionic bonds between the silver and nitrate ions. This result is completely consistent with the given answers, as we have to break strong ionic bonds between silver ion and nitrate.
What is the reaction of silver with water?
Silver does not react with pure water. Is is stable in both water and air. Moreover, it is acid and base resistant, but it corrodes when it comes in contact with sulphur compounds. Under normal conditions silver is water insoluble.
Is agno2 soluble in water?
Silver nitrite is an inorganic compound with the formula AgNO2….Silver nitrite.
| Names | |
|---|---|
| Solubility in water | 0.155 g/100 mL (0 °C) 0.275 g/100 mL (15 °C) 1.363 g/100 mL (60 °C) |
| Solubility | insoluble in ethanol |
| Magnetic susceptibility (χ) | −42.0·10−6 cm3/mol |
| Hazards |
What is the reaction of AgNO3?
Silver nitrate decomposes when heated: 2 AgNO3(l) → 2 Ag(s) + O2(g) + 2 NO2(g) Qualitatively, decomposition is negligible below the melting point, but becomes appreciable around 250 °C and fully decomposes at 440 °C.
What does silver nitrate do to the skin?
Silver nitrate topical (for use on the skin) is used to cauterize infected tissues around a skin wound. Silver nitrate can also help create a scab to help stop bleeding from a minor skin wound. Silver nitrate is also used to help remove warts or skin tags.
Does hot water react with silver?
It also starts floating due to the bubbles of hydrogen gas sticking to its surface. Metals like aluminium, iron and zinc do not react either with cold or hot water. But they react with steam to form the metal oxide and hydrogen. Metals such as lead, copper, silver and gold do not react with water at all.
What is the symbol of nitrite?
NO2−
Nitrite/Formula
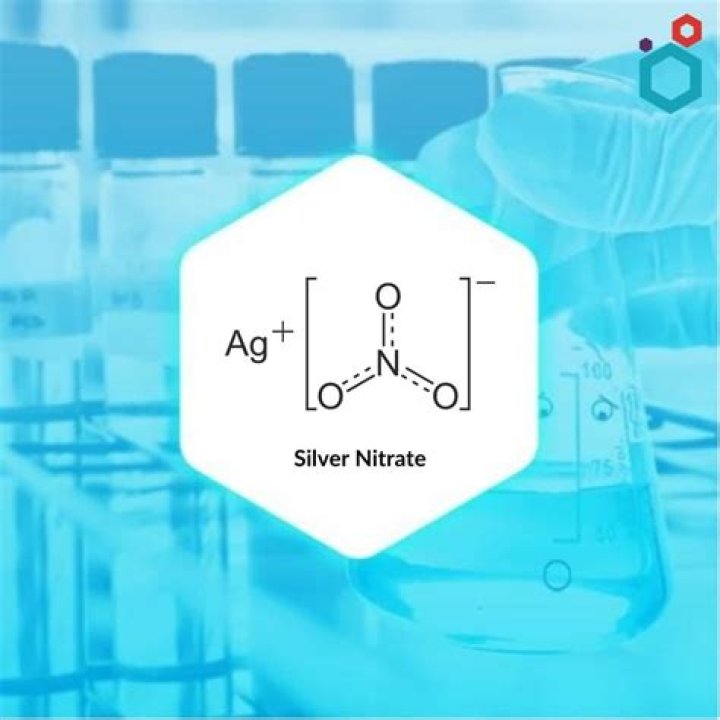
What is the formula of silver nitrate?
AgNO3
Silver nitrate/Formula
What happens to silver nitrate in pure water?
There is no reaction between the two. Silver nitrate simply dissolves in pure water. If, however, you have any chloride or other halide in water, silver nitrate reacts with that to form an insoluble precipitate of silver chloride or the respective halide.
What is the reaction between silver nitrate and ethanol?
The hazards of AgNO 3 include its toxic and corrosive nature. The reaction between silver nitrate and ethanol is explosive. The silver present in this compound is displaced by copper, which forms copper nitrate. The chemical equation for this reaction is given by 2AgNO 3 + Cu → Cu(NO 3) 2 + 2Ag.
How is silver nitrate used as an antiseptic?
Silver nitrate is a corrosive metal which is administered either in the form of a liquid or a stick form diluted with water. Silver nitrate is diluted with water in the liquid form with 0.01% to 10% concentration. This solution serves as a mild antiseptic in medical setups.
What happens when silver nitrate is added to a halogenoalkane?
Background In this case, various halogenoalkanes are treated with a solution of silver nitrate in a mixture of ethanol and water. Nothing else is added. After varying lengths of time precipitates appear as halide ions (produced from reactions of the halogenoalkanes) react with the silver ions present.