How did the scattering of alpha particles by a thin foil?
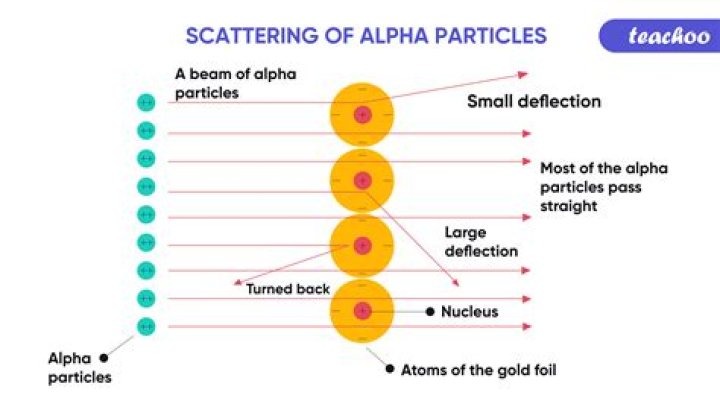
The atom being mostly empty space. A small number of alpha particles were deflected by large angles (> 4°) as they passed through the foil. There is a concentration of positive charge in the atom. Like charges repel, so the positive alpha particles were being repelled by positive charges.
What did the alpha particle scattering experiment prove?
Rutherford’s alpha scattering experiment showed that the majority of alpha particles fired at a thin sheet of gold leaf passed straight through. Some of the particles passed through the leaf with a small angle of deflection and very few were deflected at very large angles.
What will happen when the alpha particles reach the foil?
Q: What would you expect to happen when the alpha particles strike the gold foil? A: The alpha particles would penetrate the gold foil. Alpha particles are positive, so they might be repelled by any areas of positive charge inside the gold atoms.
What are alpha particle scattering?
the scattering of an alpha particle through a large angle with respect to the original direction of motion of the particle, caused by an atom (Rutherford atom ) with most of the mass and all of the positive electric charge concentrated at a center or nucleus. Also called al.
What was the purpose of Rutherford’s scattering experiment?
Rutherford’s gold foil experiment demonstrated that almost all of the mass of an atom is in a tiny volume in the center of the atom which Rutherford called the nucleus. This positively charged mass was responsible for deflecting alpha particles propelled through the gold foil.
What is the basic principle of alpha particle scattering method?
“Repulsion between the positively charged nucleus and positively charged Alpha Particle”. When alpha particles are fired towards the nucleus, due to same nature of charge they get deviated from their original path.
What kind of foil is used for alpha particle scattering?
Both scientists performed an experiment whose schematic diagram is shown in the figure given. They directed a beam of 5.5 MeV α-particles emitted from a radioactive source at a thin metal foil made of gold. The beam was allowed to fall on a thin foil of gold thickness 2.1 × 10 -7 m.
How did Rutherford demonstrate the scattering of alpha particles by gold foil?
To demonstrate the scattering of alpha particles by gold foil. Model for the structure of an atom had been first proposed by J.J. Thomson. Later, followed many theories however, Rutherford’s model was finally accepted as the correct nuclear model. Rutherford had shown his model with help of an experiment.
How is the scattering of alpha particles observed?
The beam was allowed to fall on a thin foil of gold thickness 2.1 × 10 -7 m. The scattered alpha-particles were observed through a rotatable detector consisting of zinc sulphide screen and a microscope. The scattered alpha-particles on striking the screen produced brief light flashes or scintillations.
How big is a gold foil for alpha particles?
They directed a beam of 5.5 MeV α-particles emitted from a radioactive source at a thin metal foil made of gold. The beam was allowed to fall on a thin foil of gold thickness 2.1 × 10 -7 m.