How do acids differ from base when dissolved in water?
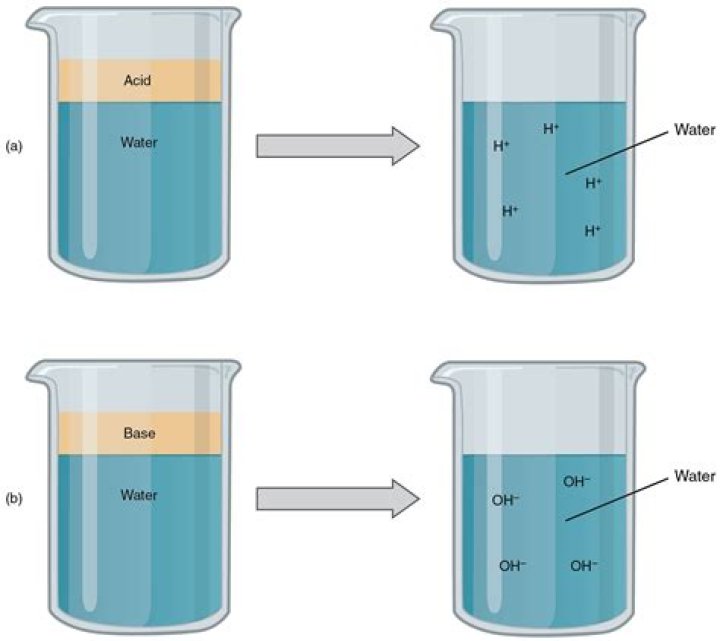
In a water solution, acids differ from bases in that acids release hydrogen ions into solution while bases release hydroxide ions into solution.
What compound do acids and bases produce when dissolved in water?
In this reaction, sodium hydroxide (NaOH) disassociates into sodium (Na+) and hydroxide (OH-) ions when dissolved in water, thereby releasing OH- ions into solution. Arrhenius acids are substances which produce hydrogen ions in solution. Arrhenius bases are substances which produce hydroxide ions in solution.
Do acids and bases produce ions when dissolved in water?
Sodium hydroxide (NaOH) is a strong base because when placed in water, it dissociates completely into sodium ions (Na+) and hydroxyl ions (OH-), all of which are now released and dissolved in water. Acids, bases and salts, dissociate (separate) into electrolytes (ions) when placed in water.
What do bases produce when dissolved in water?
hydroxide ions
Bases can be defined as “any substance that, when dissolved in water, generates hydroxide ions (OH-)”. Hydroxide ions are produced under two conditions.
Is H+ acidic or basic?
Now there are more hydrogen ions than hydroxide ions in the solution. This kind of solution is acidic. A base is a substance that accepts hydrogen ions….What does it mean for a solution to be acidic or basic (alkaline)?
| pH Value | H+ Concentration Relative to Pure Water | Example |
|---|---|---|
| 1 | 1 000 000 | gastric acid |
What happens to acid or base in water solution?
Acid and bases in water Acids and bases dissolve in water and, because they increase the concentration of either protons or hydroxide ions, they suppress water self-ionization. Acids in water solution dissociate H+ ions. Base, when dissolved in water, produces OH– ion.
Is OH a base or acid?
OH – is called a hydroxyl ion and it makes things basic. However, in water, there is a balance between hydroniums and hydroxyls so they cancel each others’ charges. Pure water is neither acidic or basic; it is neutral. So how does something become acidic or basic?
What happens if base is dissolved in water explain with an example?
When a base is is dissolved in water dissociates to form a conjugate acid and conjugate base. The conjugate base and acid are ions. The conjugate base is stronger than the conjugate acid.
Is oh a base or acid?
Which is ion is produced when a base is dissolved in water?
E.g ammonia, NH3, acts as a Bronsted-Lowry base by accepting a H+ ion in the reaction NH3 + H^+ →NH4+. This is a slight simplification of the real reaction, where the H+ ion bonds to a H2O molecule first, but point is bases which are not hydroxides usually react directly with acids.
What makes an acid dissociate from a base?
Depends on the concentration of hydroxide ions. Acids dissociate to give free hydrogen ions (H+) after mixing in water. Bases dissociate to give free hydroxide ions (OH-) after mixing in water. Hydrochloric acid (HCl), Sulphuric acid (H2SO4), Nitric acid (HNO3), Carbonic acid (H2CO3).
What does an acid produce when it is dissolved in water?
Most acids release H + ions in water, which combines with water molecule to produce hydronium ( H 3 O +) ion. Eg. But some compounds show acidic nature in water, but don’t have any Hydrogen attached to them. Such compounds are Lewis acids i.e. they accept lone pair of electrons from doner atom.