How do you make a gold chloride solution?
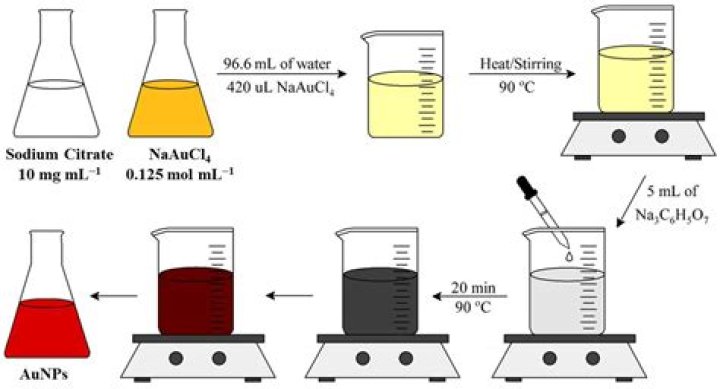
1. Preparation of Reagents
- Preparation of gold chloride. Dissolve 1 g of gold(III) chloride trihydrate in 100 g of H2O to give 25 mM HAuCl4.
- Preparation of borax (Na2B4O7·10H2O)
- Preparation of sodium thiocyanate.
- Preparation of sodium carbonate.
- Preparation of glutathione.
How much gold is gold chloride?
Gold Chloride 100ml of 5% solution contains exactly 5.00 grams of pure Gold Metal. Contains 5.000 grams of 99.99% pure gold ions in De-Ionized water.
How do you make a chemical gold?
Gold is the chemical element with 79 protons in each atomic nucleus. Every atom containing 79 protons is a gold atom, and all gold atoms behave the same chemically. In principle, we can therefore create gold by simply assembling 79 protons (and enough neutrons to make the nucleus stable).
Is gold chloride soluble in water?
Gold(III) chloride is very hygroscopic and highly soluble in water and ethanol. It decomposes above 160 °C or in light, and it forms a variety of complexes with many ligands.
What is gold chloride used for?
a yellow to red, water-soluble compound, AuCl3, used chiefly in photography, gilding ceramic ware and glass, and in the manufacture of purple of Cassius. Also called gold trichloride.
Does gold chloride dissolve in water?
Gold(III) chloride
| Names | |
|---|---|
| Melting point | 254 °C (489 °F; 527 K) (decomposes) |
| Solubility in water | 68 g/100 ml (cold) |
| Solubility | soluble in ether and ethanol, slightly soluble in liquid ammonia |
| Magnetic susceptibility (χ) | −112·10−6 cm3/mol |
Where is gold chloride found?
The compounds gold chloride (AuCl) and hydrogen gold chloride (HAuCl2) play important natural roles in making gold accessible to us. Steamed out of Magma. Volcanoes can be thought of as places on Earth where melted rock from deep underground has squeezed out upon the surface and mounded up, forming volcanic mountains.
Can we turn lead into gold?
Nuclear Transmutation. In modern times, it has been discovered that lead can in fact be turned into gold, but not through alchemy, and only in insignificant amounts. Nuclear transmutation involves the use of a particle accelerator to change one element into another.
How long does it take to prepare gold chloride?
Gold Monochloride or Aurous Gold Chloride “AuCl” is a salt is prepared by heating the trichloride to 185° in air for twelve hours.
Is it possible to turn metal into gold?
Yes, surely in principle. Why into gold, you can convert one metal to any other metal simply finding a method of changing the number of neutrons and protons in the nucleus. Clearly what differentiates two elements is the different number of neutrons and protons- so named as atomic weight.
What do you need to make gold plating solution?
Step 3: Final Step, Making Gold Plating Solution. In the final step, we need to make a strongly alkaline solution of gold chloride and potassium ferrocyanide. Using this solution and electrical current, we can deposit gold over the surface of other metals. This solution contains gold cyanide ions, which are extremly poisonous.
When does gold chloride turn into hydrogen peroxide?
A similar change, yielding hydrogen peroxide, was found by Sonstadt to take place when solutions containing 0.04 per cent, and also 0.007 per cent, of gold chloride were exposed to bright sunshine for several days. The presence of free hydrochloric acid prevents this decomposition.