How do you separate metals from rocks?
The rock is then crushed by powerful machinery. Metal is extracted from the crushed ore by one of two major methods: smelting or electrolysis. Smelting uses heat to separate the valuable metal from the rest of the ore. Smelting usually requires a reduction agent, or another chemical, to separate metal from its ore.
What is the process of separating metals from their ores called?
Extractive metallurgy is the practice of removing valuable metals from an ore and refining the extracted raw metals into a purer form. Hydrometallurgy uses aqueous solutions to extract metals from ores (leaching). Pyrometallurgy involves high temperature processes where chemical reactions take place.
How are metal separated?
A metal is separated from its ore and then isolated by using pyrometallurgy or hydrometallurgy or by taking advantage of unusual chemical or physical properties of a particular compound.
What physical and chemical properties are used to separate metals from their ores?
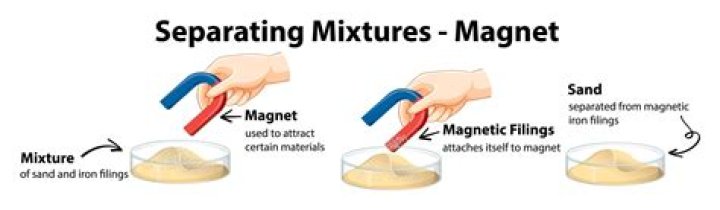
Major physical and chemical methods include magnetic separation, froth flotation, leaching etc., whereby the impurities and unwanted materials are removed from the ore and the base ore of the metal is concentrated, meaning the percentage of metal in the ore is increased.
Which metal can be purified by Liquation?
Lead , tin and bismuth are purified by liquation method.
How do you separate copper and rock?
Copper can be extracted from its ore by: Underground: sinking a vertical shaft into the Earth to an appropriate depth and driving horizontal tunnels into the ore. Open pit: 90% of ore is mined by this method. Ores near the surface can be quarried after removal of the surface layers.
How do you separate impurities from metals?
These methods include: Distillation, which involves vaporizing the metal and then allowing it to solidify outside of the impurities. Liquation, which involves melting the metal, and allowing it to run out of the solid impurities.
How do you separate minerals from its ore?
A successful separation of a valuable mineral from its ore can be determined by heavy-liquid testing, in which a single-sized fraction of a ground ore is suspended in a liquid of high specific gravity. Particles of less density than the liquid remain afloat, while denser particles sink.
Can you separate metals?
Liquation is a metallurgical method for separating metals from an ore or alloy. A mixture of metals is melted together with a third, after which the mixture is separated by liquid extraction.
Is gold not rusting a physical property?
Some examples of chemical properties are: paper burns. iron rusts. gold does not rust.
What’s the process of separating metal from liquid?
Adsorption, a process of separating out metal solids from liquid solutions, is common, as is smelting, a process that removes different types of metals from one another.
What is the process of separating minerals from their ores?
In the field of extractive metallurgy, mineral processing, also known as ore dressing, is the process of separating commercially valuable minerals from their ores . This section needs expansion. You can help by adding to it. (August 2010)
How are metals extracted from the waste rock?
1) or “native” metals (such as native copper) that are not commonly concentrated in the Earth’s crust, or “noble” metals (not usually forming compounds) such as gold (Figure 23.2. 2 ). The ores must be processed to extract the metals of interest from the waste rock and from the ore minerals.
Do you need to know how to separate metals?
In order to recover those metals once the object has worn out or broken, you’ll need to properly know how to separate the metal. Adsorption, a process of separating out metal solids from liquid solutions, is common, as is smelting, a process that removes different types of metals from one another.