How do you tell if an element will lose or gain electrons?
In general, metals will lose electrons to become a positive cation and nonmetals will gain electrons to become a negative anion. Hydrogen is an exception, as it will usually lose its electron.
What metals lose or gain electrons?
Metals tend to lose electrons and non-metals tend to gain electrons, so in reactions involving these two groups, there is electron transfer from the metal to the non-metal. The metal is oxidized and the non-metal is reduced. An example of this is the reaction between the metal, sodium, and the non-metal, chlorine.
Does gold gain electrons in chemical changes?
Chemically, gold is a transition metal and a group 11 element.As it is a metal it loses the electrons during a chemical change.
Can objects lose or gain electrons?
For an object to become charged, it must either gain or lose electrons. Losing electrons results in more positive charge than negative charge, making the object charged positively. Gaining electrons results in more negative charge than positive charge, making the object charged negatively.
How many electrons are gained or lost in nitrogen?
3 electrons
But typically a nitrogen atom gains 3 electrons to form the nitride ion, N3− .
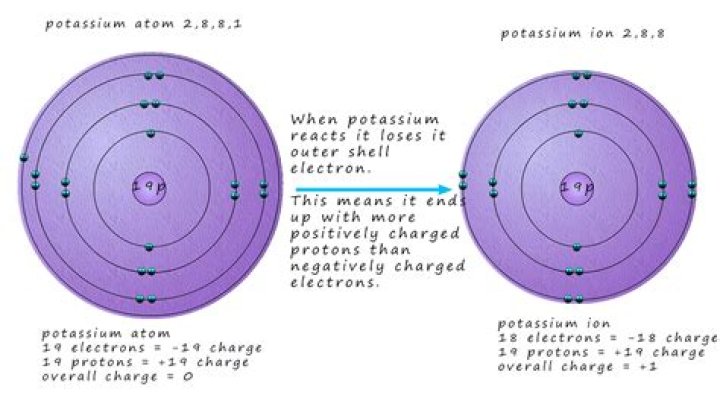
Which element loses an electron most easily?
Potassium is most likely to lose an electron.
What gains electrons more easily?
Answer: Among all the elements, hydrogen gains 1 electron to complete their duplet. Sodium (na), potassium (k), magnesium (mg) all losses their electron to attain stability as noble gas.
Which element is most likely to gain electrons?
Nonmetals
Nonmetals, which are found in the right-hand region of the periodic table, have relatively large ionization energies and therefore tend to gain electrons.
Which elements would expect to gain electrons?
So, among the given options, nitrogen, and iodine can gain electrons. All other elements mentioned are metals, donate electrons, and get converted into cations.
Does sulfur lose electrons in a chemical reaction?
Atomic number of sulphur = 16 In order to attain noble gas configuration, it has to looses 6 electrons which is not favorable. So, sulphur gains electrons instead of loosing.