How is strength of acids and base determined?
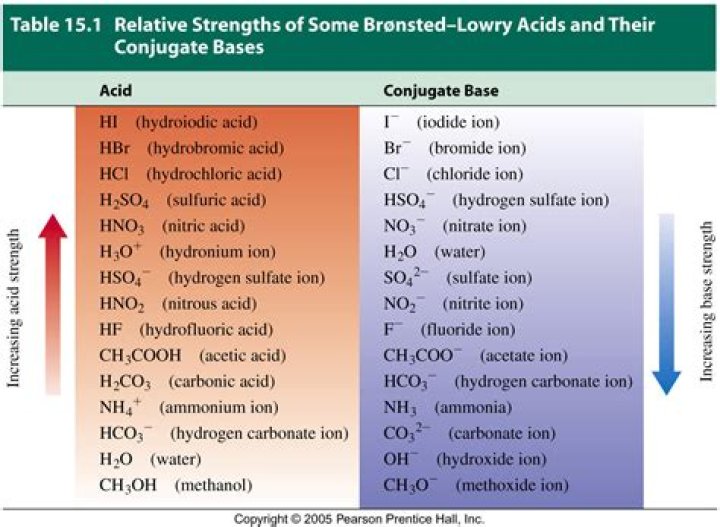
Acids and bases fall into the category of being either weak or strong bases on the following criteria. The acid and base dissociation constant is the measure of the strength of the acids and bases. The higher the dissociation constant the stronger the acid or base. Acids and bases are measured using the pH scale.
Which of the following is the strongest base ABCD?
-Hence Benzyl amine is the strongest base among given compounds. So, the correct answer is “Option D”.
Is LiOH a weak base?
LiOH is a weak base because it is more covalent due to small size. So, they cannot yield hydroxyl ions easily in aqueous solutions which is the main cause of basicity.
What are the 7 strong bases?
Strong Arrhenius Bases
- Potassium hydroxide (KOH)
- Sodium hydroxide (NaOH)
- Barium hydroxide (Ba(OH)2)
- Caesium hydroxide (CsOH)
- Strontium hydroxide (Sr(OH)2)
- Calcium hydroxide (Ca(OH)2)
- Lithium hydroxide (LiOH)
- Rubidium hydroxide (RbOH)
Which of the following is the the strongest base?
Benzylamine (C6H5CH2NH2) is the strongest base among all because the aliphatic amines are more basic than aromatic amines.
What determines a strong base and a weak base?
What determines a Strong Base and a Weak Base Strong Bases and Weak Bases : The strength of a base is determined by the amount of hydroxide ions (OH –) that the base provides when dissolved in water. Some of the bases, when dissolved in water, get almost completely dissociated to provide hydroxide ions.
How to determine the strength of an acid?
You should commit the strong acids to memory: 1 HCl: hydrochloric acid 2 HNO 3: nitric acid 3 H 2 SO 4: sulfuric acid 4 HBr: hydrobromic acid 5 HI: hydroiodic acid 6 HClO 4: perchloric acid
How are strong acids and weak bases dissociated in water?
Strong electrolytes are completely dissociated into ions in water. The acid or base molecule does not exist in aqueous solution, only ions. Weak electrolytes are incompletely dissociated. Here are definitions and examples of strong and weak acids and strong and weak bases. Strong acids completely dissociate in water, forming H + and an anion.
Which is a strong base in an organic acid?
Organic acids containing one or more carboxyl group, -COOH. The H is ionizable. Anions with an ionizable proton (e.g., HSO 4- → H + + SO 42- ). Strong bases dissociate 100 percent into the cation and OH – (hydroxide ion). The hydroxides of the Group I and Group II metals usually are considered to be strong bases.