How many electron are there in gold?
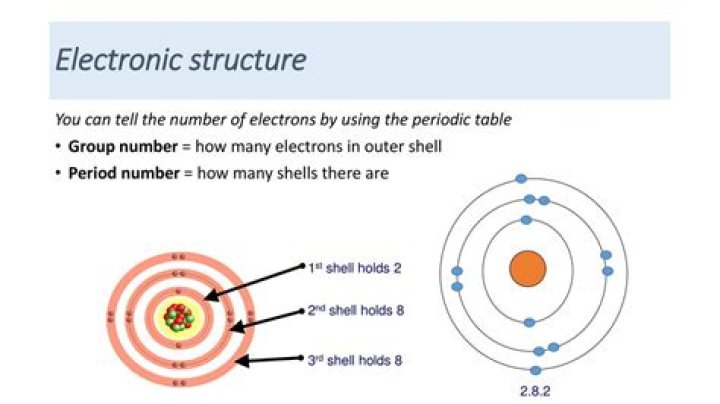
79
| Name | Gold |
|---|---|
| Number of Protons | 79 |
| Number of Neutrons | 118 |
| Number of Electrons | 79 |
| Melting Point | 1064.43° C |
Does gold have free electrons?
Silver, iron, platinum, gold, and copper are all metals, which generally are malleable and ductile, conduct electricity and heat, and have a metallic luster. The electrons are free to move throughout the metal, which explains its high electrical and thermal conductivities and luster.
Are there atoms in gold?
All regular matter is made out of atoms. Gold is the chemical element with 79 protons in each atomic nucleus. Every atom containing 79 protons is a gold atom, and all gold atoms behave the same chemically.
Where are the electrons located in the gold?
How many electrons are in gold? Electrons are negatively charged particles with an extremely small mass. They are located in the electron cloud of atoms. Electrons can be added or removed from an atom without changing what that atom is, but it does change the electrical charge of the atom. See full answer below.
What’s the atomic number of the element gold?
Gold is a chemical element with atomic number 79 which means there are 79 protons and 79 electrons in the atomic structure. The chemical symbol for Gold is Au. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons.
Where does gold come from in the periodic table?
Gold is thought to have been produced in supernova nucleosynthesis, from the collision of neutron stars. Gold is a chemical element with atomic number 79 which means there are 79 protons in its nucleus.
What is the electronegativity of the element gold?
Electronegativity of Gold is 2.54. Electronegativity, symbol χ, is a chemical property that describes the tendency of an atom to attract electrons towards this atom. For this purposes, a dimensionless quantity the Pauling scale, symbol χ, is the most commonly used.