How many electron does Mercury have?
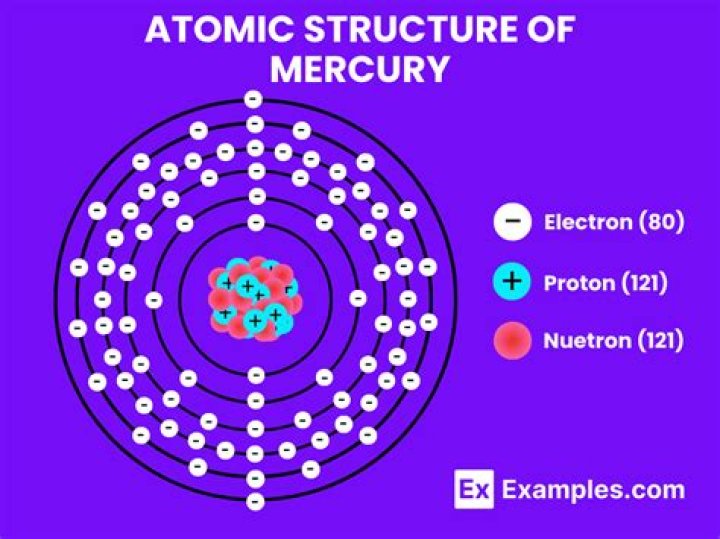
80
| Name | Mercury |
|---|---|
| Atomic Mass | 200.59 atomic mass units |
| Number of Protons | 80 |
| Number of Neutrons | 121 |
| Number of Electrons | 80 |
How many electron does gold have?
79
| Name | Gold |
|---|---|
| Number of Protons | 79 |
| Number of Neutrons | 118 |
| Number of Electrons | 79 |
| Melting Point | 1064.43° C |
Does gold have more protons in mercury?
Usually gold is created from platinum, which has one less proton than gold, or from mercury, which has one more proton than gold.
What is the smallest particle of mercury?
electron O molecule atom proton.
Is mercury a heavier element than tin?
Yes, mercury is heavier than tin. Mercury and tin are both elements. To find which is heavier, you need to look at the periodic table. As the atomic…
Can you turn mercury into gold?
Gold was synthesized from mercury by neutron bombardment in 1941, but the isotopes of gold produced were all radioactive. Using fast neutrons, the mercury isotope 198Hg, which composes 9.97% of natural mercury, can be converted by splitting off a neutron and becoming 197Hg, which then decays into stable gold.
Where is mercury found naturally?
What is Mercury? Mercury is a naturally-occurring chemical element found in rock in the earth’s crust, including in deposits of coal.
How much mercury does it take to make one ounce of gold?
Mercury is sometimes added direct to the outside plates, and sometimes their condition is regulated by the additions of mercury in the mortar box. The amount added varies with the conditions of crushing and the richness of the ore, but in general from 1 to 2 ounces of mercury are fed in for every ounce of gold contained in the ore.
Which is heavier an atom of gold or mercury?
An atom of gold is lighter than an atom of lead; however, gold is more dense than mercury, so a cubic centimeter of gold is heavier than a cubic centimeter of mercury.
How many electrons and protons does Mercury have?
Mercury atoms have 80 electrons and 80 protons with 122 neutrons in the most abundant isotope. …
How are the electrons of gold listed on the periodic table?
The number of electrons in each element’s electron shells, particularly the outermost valence shell, is the primary factor in determining its chemical bonding behavior. In the periodic table, the elements are listed in order of increasing atomic number Z. Electron configuration of Gold is [Xe] 4f14 5d10 6s1. Possible oxidation states are +1,3.