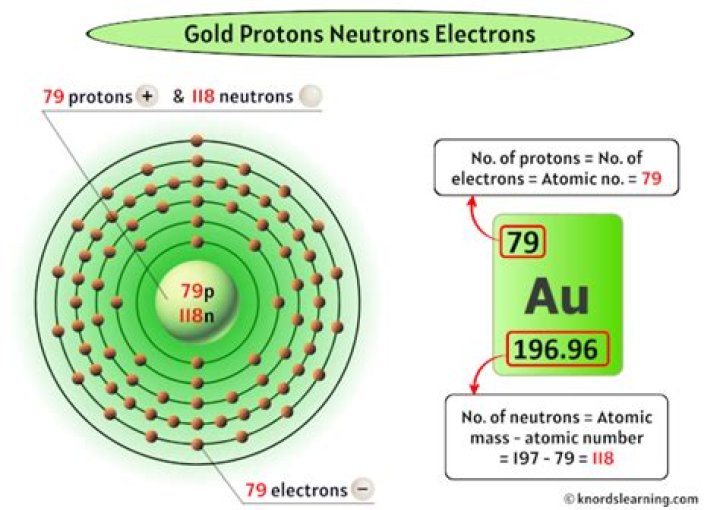
How many protons neutrons and electrons does an atom of gold have?
| Name | Gold |
|---|---|
| Atomic Mass | 196.967 atomic mass units |
| Number of Protons | 79 |
| Number of Neutrons | 118 |
| Number of Electrons | 79 |
What kind of structure is gold?
| Gold | |
|---|---|
| Spectral lines of gold | |
| Other properties | |
| Natural occurrence | primordial |
| Crystal structure | face-centered cubic (fcc) |
What is the electron charge of gold?
Gold also has 79 negatively-charged electrons; these will balance the 79 positively-charged protons. These electrons will exist in determined orbitals around the nucleus.
How many neutrons are in a gold atom, and why?
The ratio Z/N deviates from 1:1! For gold it is 1 proton per 1.5 neutrons! For the heaviest elements beyond lead, even an enormous surplus of neutrons cannot stabilize them any longer and they decay as a consequence. The reason is: The strong nuclear force that keeps the core together is very…
How many protons and neutrons are in an atom?
The atomic number (Z): The number of protons or the number of electrons present in an atom is known as atomic number. Mass number (A): The total number of protons and neutrons present in the nucleus of an atom is known as mass number. For Mg, Atomic number is 12 and the mass number is 24.
How many protons does aluminium have in its nucleus?
Since you’re dealing with an isotope of aluminium, it follows that this atom must have the exact same number of protons in its nucleus. The number of protons an atom has in its nucleus is given by the atomic number. A quick looks in the periodic table will show that aluminium has an atomic number equal to 13.
How many electrons and neutrons are in the element sodium?
The element sodium has 12 neutrons, 11 electrons and 11 protons. The number of electrons and protons come from the element’s atomic number, which is same 11. The number of neutrons can be found by subtraction of the atomic number from sodium’s atomic mass of twenty three. Likewise, how many protons electrons and neutrons are in a given element?