Is melting a physical change or a chemical change explain your answer?
Melting is a physical change because it involves the changing of matter from a solid state into a liquid one, without any change in the chemical…
Why is melting a chemical change?
Melting is the phase change in the state of matter from solid to liquid. When something melts, it doesn’t change chemically, so melting is not a…
Is melting and boiling a physical or chemical change?
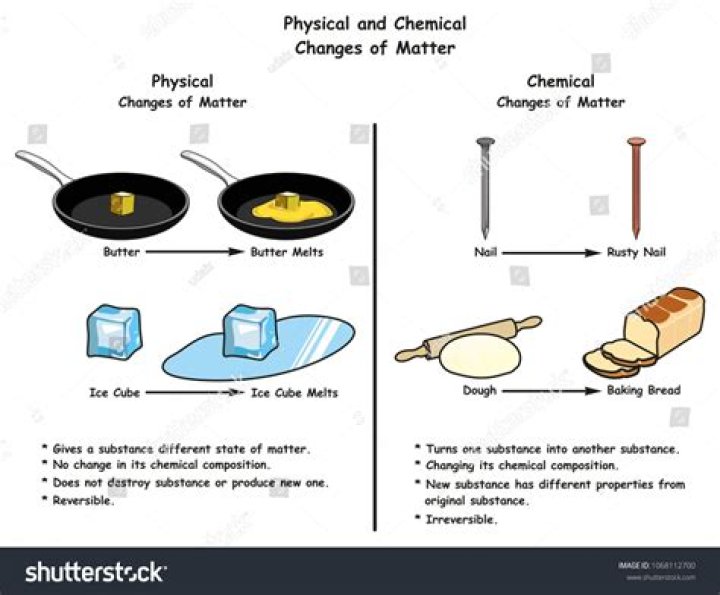
Physical changes alter only the size, shape, form or matter state of a material. Water boiling, melting ice, tearing paper, freezing water and crushing a can are all examples of physical changes.
What are 4 examples of physical change?
Examples of Physical Changes
- Crushing a can.
- Melting an ice cube.
- Boiling water.
- Mixing sand and water.
- Breaking a glass.
- Dissolving sugar and water.
- Shredding paper.
- Chopping wood.
What are two major types of physical change?
Physical changes affect a substance’s physical properties but do not alter its chemical structure. Types of physical changes include boiling, clouding, dissolution, freezing, freeze-drying, frost, liquefaction, melting, smoke and vaporization.
Is melting a crayon a chemical change?
If a crayon was melting in the sun it would be a Chemical change!!
Is melting plastic a chemical change?
Melting plastic is not a chemical reaction, but rather is a physical change. When a substance changes its phase, such as solid melting to liquid, it…
Is heat needed in physical and chemical change?
Application of heat to certain substances causes only physical changes in which no new substance or substances are formed. Application of heat to some substances causes chemical changes, or chemical reactions, in which one or more new substances are formed, with different properties from the original.
What’s the difference between melting and a physical change?
Melting is a physical change, not a physical property. The distinction is subtle, but the way I think of it is: A physical change is one that does not involve a change in any chemical bonds.
Is the melting of gold a physical property?
It is a phase change – gold exists as a solid crystal (fcc I believe off the top of my head), as a liquid, and as a vapor (gas). First order phase transitions exhibit a volume change, and an enthalpy of the phase change. In the case of gold, both the crystal and the liquid are metallic, with similar numbers of nearest neighbors.
How are physical changes and chemical changes reversible?
Many physical changes are reversible, if sufficient energy is supplied. The only way to reverse a chemical change is via another chemical reaction. Examples of Chemical Changes
Is the melting of butter a physical change or chemical change?
If your talking about butter slightly melting on a hot day because it was left out on the counter, THAT is a physical change and is reversible by simply putting the butter into the fridge.