Is Neon a solid liquid or gas at room temperature?
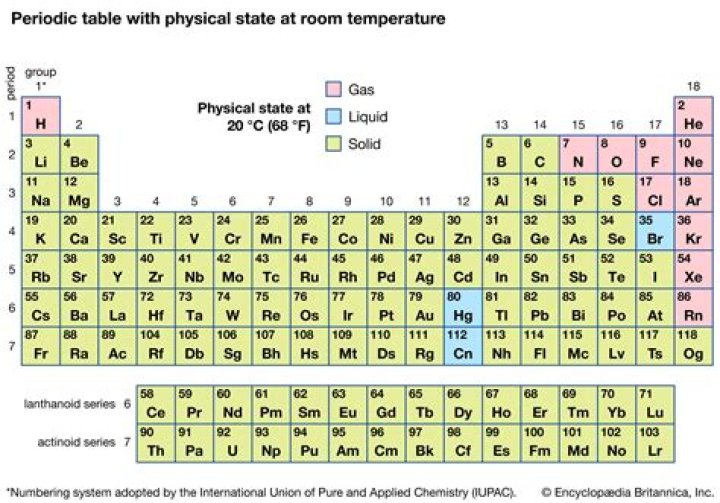
Classified as a noble gas, Neon is a gas at room temperature.
Which elements are solid at room temperature?
Elements that are solids at room temperature include sodium, antimony, gold, silver and platinum. Other such elements are arsenic, calcium, carbon, boron and tungsten. Iron, lead, palladium and tin are also solid at room temperature.
Is aluminum a solid liquid or gas?
Aluminum is a solid at room temperature.
Which metal is gas at room temperature?
Eleven non-metals are gases at room temperature, including oxygen and chlorine. One non-metal, bromine, is a liquid at room temperature. The other non-metals are solids at room temperature, including carbon and sulfur.
Is shiny is solid at room temperature?
Metals: Substances that are hard solid at room temperature (except mercury), shiny, malleable, ductile, and good conductor of heat and electricity. Non-Metals: Substances that are gases or brittle solids at room temperature, not shiny, not malleable, not ductile, and poor conductors of heat and electricity.
Is CO2 solid at room temperature?
Carbon dioxide is a gas at room temperature while silicon dioxide is a solid at room temperature with a melting point of 1770°C.
Is aluminium solid at room temperature?
Aluminum is a chemical element with symbol Al and atomic number 13. Classified as a post-transition metal, Aluminum is a solid at room temperature.
What are two gas examples?
Examples of Gases
- Hydrogen.
- Nitrogen.
- Oxygen.
- Carbon Dioxide.
- Carbon Monoxide.
- Water Vapour.
- Helium.
- Neon.
Which is less radioactive depleted uranium or natural uranium?
Depleted uranium is uranium that has much less uranium-235 than natural uranium. It is considerably less radioactive than natural uranium. It is a dense metal that can be used as ballast for ships and counterweights for aircraft.
Why does uranium have a wide range of oxidation states?
This means that a variety of orbitals can now be combined to make bonds, and from this, some very interesting compounds. In the absence of air, uranium can display a wide range of oxidation states, unlike the lanthanides just above it, and it forms many deeply coloured complexes in its lower oxidation states.
How did uranium get its name on the periodic table?
Uranium was named after the planet Uranus. A vertical column in the periodic table. Members of a group typically have similar properties and electron configurations in their outer shell. A horizontal row in the periodic table. The atomic number of each element increases by one, reading from left to right.
Why does uranium have so many protons and neutrons?
Chemically, uranium is fascinating. Its nucleus is so full of protons and neutrons that it draws its core electron shells in close. This means relativistic effects come into play that affect the electron orbital energies. The inner core s electrons move faster, and are drawn in to the heavy nucleus, shielding it better.