Is tarnished silver a chemical change?
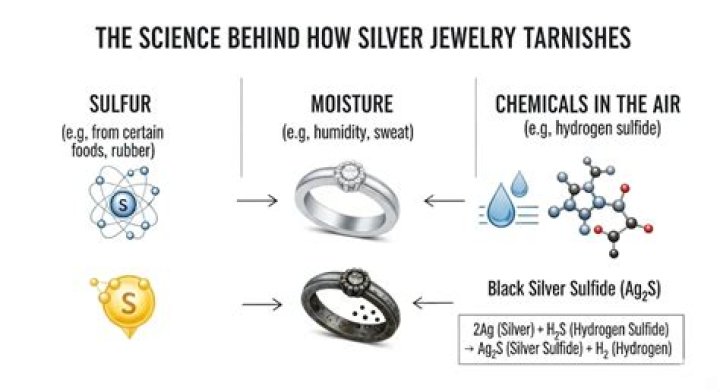
Yes, tarnishing of silver metal is a chemical change, as opposed to a physical change. Silver metal reacts with sulfur compounds (including minute amounts of Hydrogen Sulfide in the atmosphere) to form Silver Sulfide.
Does silver Tarnish protect the silver from further corrosion?
Silver is known in the chemistry world as a noble metal which means it is resistant to corrosion, but not completely. Whether silver plating or pure silver, the composite of the metal will tarnish when exposed to air and sulfur. The original silver surface can be restored if the layer of silver sulfide is removed.
What is tarnish in chemistry?
Tarnish is the product of a chemical reaction between a metal and a non-metal compound such as oxygen or sulfur dioxide. A thin layer of corrosion forms over the metal, which mars its shine.
How do you remove heavy silver tarnish?
How to Clean Large Silver Items:
- Line your sink with foil.
- Pour boiling water into the sink.
- Add 1 cup baking soda and 1 cup salt to the water.
- Place silver pieces in the solution.
- Allow pieces to soak for up to 30 minutes.
- Remove items when cool and dry them off with a soft cloth.
What is the formula of corroded silver?
Silver objects corrode slowly in the air due to the presence of H2S in the air. The chemical equation of this reaction is 2Ag + O2 = Ag2O.
What is tarnishing give an example?
Tarnish is defined as to spoil or to discolor the surface of a piece of metal. An example of to tarnish is to expose silver to sulfur and air. To dull the luster of; discolor, especially by exposure to air or dirt. Being in the ground for so long tarnished the old coins.
What is the chemical equation for silver tarnish?
The chemical equation for the formation of silver tarnish following reaction with hydrogen sulphide (H 2 S) is as follows:
Which is the best silver anti tarnish chemical?
We are one of the major companies in the industry offering some of the finest quality Silver Anti Tarnish Chemical. These products are easy to use and deliver an even coating on the treated surface to protect it from corrosion and damage by moisture.
How do you remove silver tarnish from air?
Removing Tarnish from Silver. Silver tarnishes in air to form a black coating of silver sulphide, which has the formula Ag2S. The chemical equation for the formation of silver tarnish following reaction with hydrogen sulphide (H2S) is as follows: 2 Ag(s) + H2S(g) → Ag2S(s) + H2(g)
Why does sterling silver have a bad tarnish?
So why does silver tarnish? Sterling silver is an alloy of silver and base metals such as copper; the copper reacts with sulphur-containing gases in the air to form a layer of the compound copper sulphide (Cu2S). Pure silver is less prone to tarnish, but will still react to form silver sulphide (Ag2S).