What are the observations of Rutherford gold foil experiment?
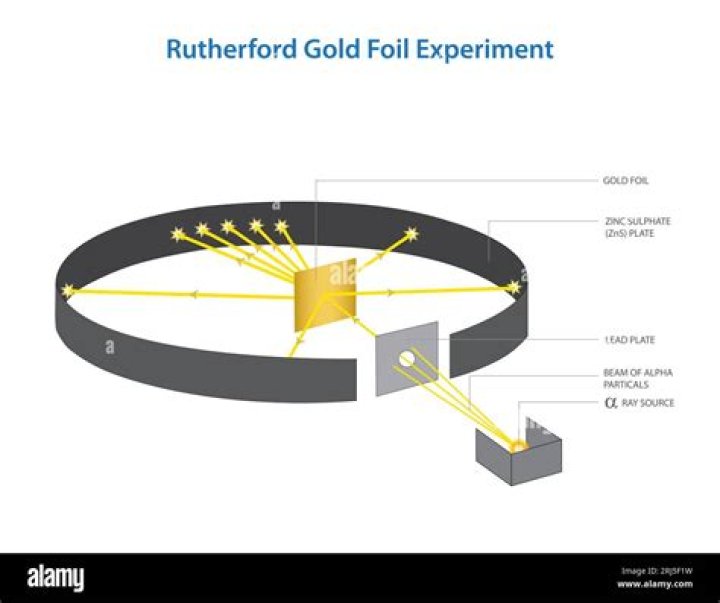
His two primary observations were: Most α particles passed straight through the gold foil, which showed that atoms are mostly empty space. Some of the α particles were deflected at various angles, and sometimes even back at the radioactive source.
What are the observations and conclusions of Rutherford’s gold foil experiment?
Rutherford considered these observations and he concluded: The fact that most alpha particles went straight through the foil is evidence for the atom being mostly empty space. A small number of alpha particles being deflected at large angles suggested that there is a concentration of positive charge in the atom.
What are the observation made by Rutherford?
The observations made by Rutherford in his scattering experiment are as follows: He observed that most of the alpha particles passed straight through the gold foil. Some were deflected through small angles and some were deflected through large angles. Very few appeared to bounce back.
What were the two main conclusions of the gold foil experiment?
From the location and number of α-particles reaching the screen, Rutherford concluded the following: i) Almost 99% of the α-particles pass through the gold foil without any deflection. So atom must be having a lot of empty space in it. ii) Several α-particles get deflected at angles.
Why was the gold-foil experiment important?
Rutherford’s experiment showed that atoms consisted of a dense mass which was surrounded by mostly empty space – the nucleus! The conclusion that could be formed from this result was that atoms had an inner core which contained most of the mass of an atom and was positively charged.
What was the purpose of the gold foil experiment?
Rutherford’s gold foil experiment (Rutherford’s alpha particle scattering experiment) refers to an experiment carried out by Ernest Rutherford, Hans Geiger, and Ernest Marsden at the University of Manchester in the early 1900s. In the experiment, Rutherford and his two students studied how alpha particles fired at a thin piece …
What kind of foil is used in the Rutherford experiment?
The Rutherford Experiment. The tutorial simulates diffraction of alpha particles (helium nuclei containing two positive charges) by a thin foil made of gold metal. Opposite the gold foil is a zinc sulfide screen that emits a flash of light when struck by an alpha particle. To operate the tutorial, use the slider to increase…
What was the observations and conclusions made by Rutherford?
Explain the gold foil experiment by Rutherford. What were the observations and conclusions made by Rutherford ? Rutherford took a very thin gold foil with 100 nm thickness. He bombarded the gold foil with alpha rays. It is important to note that alpha particles are positively charged.
Why was gold used in the Rutherford experiment?
They used gold since it is highly malleable, producing sheets that can be only a few atoms thick, thereby ensuring smooth passage of the alpha particles. A circular screen coated with zinc sulfide surrounded the foil.