What are the three main observations of the gold-foil experiment?
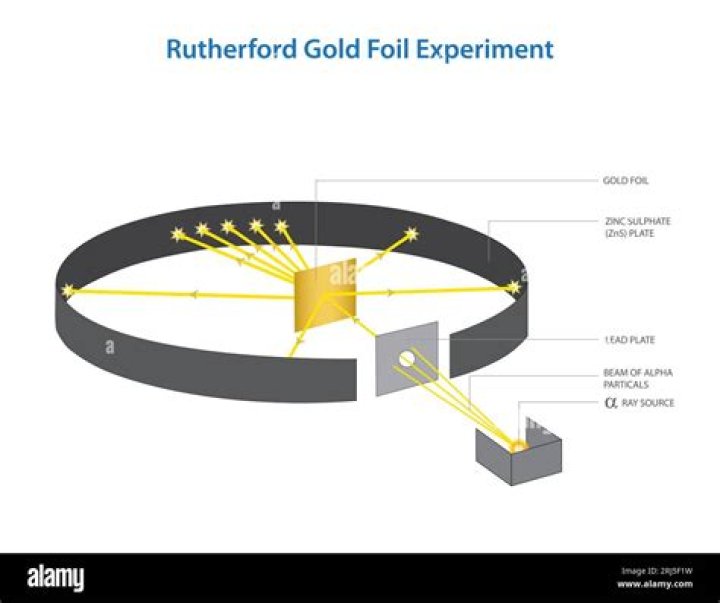
What were the three major observations Rutherford made in the gold foil experiment?
- Major fraction of alpha particles pass through the foil undeflected.
- A few alpha-particles were scattered by very small angles.
- A very small number of particles were deflected back by 1800.
What are the two main findings from the gold-foil experiment?
His two primary observations were: Most α particles passed straight through the gold foil, which showed that atoms are mostly empty space. Some of the α particles were deflected at various angles, and sometimes even back at the radioactive source.
What is gold-foil experiment in short?
Rutherford in 1911, carried out an experiment called ‘Gold foil experiment’ and could conclude the nature of an atom and the position of the protons present in the atom decisively. He also proposed the position and behaviour of electrons. They bombarded fragile sheets of gold foil with fast-moving alpha particles.
What metal was used in the gold-foil experiment?
Ernest Rutherford and the gold foil experiment In this case, Rutherford placed a sample of radium (a radioactive metal) inside a lead box with a small pinhole in it. Most of the radiation was absorbed by the lead, but a thin beam of α particles escaped out of the pinhole in the direction of the gold foil.
Why do we use gold foil?
Alpha particle scattering experiment was carried out by Rutherford in 1911 which is also known as Gold foil experiment. The reason for using gold foil was that very thin foil for the experiment was required, since gold is malleable from all other metals so it can be easily shaped into very thin sheets.
Why was the gold foil experiment important?
Rutherford’s experiment showed that atoms consisted of a dense mass which was surrounded by mostly empty space – the nucleus! The conclusion that could be formed from this result was that atoms had an inner core which contained most of the mass of an atom and was positively charged.
What kind of experiment was the gold foil experiment?
Rutherford’s gold foil experiment is also sometimes referred to as the Geiger-Marsden experiment. The gold foil experiment consisted of a series of tests in which a positively charged helium particle was shot at a very thin layer of gold foil.
Why was a very thin sheet of gold foil used?
Why was a ”very thin sheet of gold metal (i.e. gold foil)” used in Rutherford’s alpha particle scattering experiment? What was the advantage of taking such a very thin sheet of gold metal in the experiment? People who retire comfortably avoid these 7 mistakes. Financial advisors are a crucial part of retirement plans.
What kind of foil was used in Rutherford’s experiment?
In this experiment, Rutherford used Gold Foil which was extremely thin sheet, not more than 1000 atoms thick. Gold Foil was bombarded with positively charged α-particles.
How are electrons held in a gold foil atom?
Well, the electrons of the gold atom were held there by the nucleus (or the 79 protons) of the gold atom. Alpha particles only have 2 protons, so the positive charge is not strong enough to attract the electrons in the gold atom.