What determines the strength of a compound?
In general, the strength of an acid in an organic compound is directly proportional to the stability of the acid’s conjugate base. Delocalization of the negative charge (such that one atom doesn’t have to bear the full negative charge) makes the molecule more stable.
How is the strength of an acid determined?
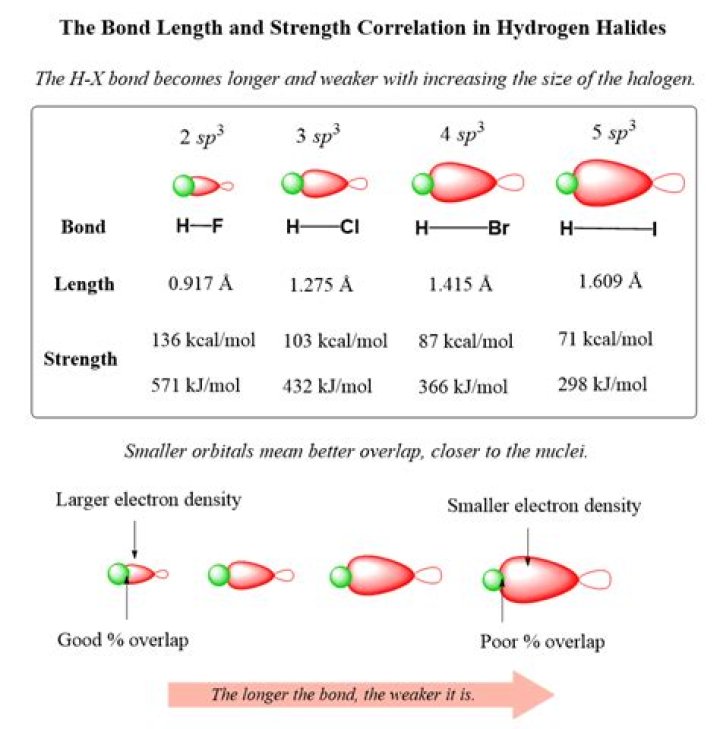
Bond Strength and Acids The bond strength of an acid generally depends on the size of the ‘A’ atom: the smaller the ‘A’ atom, the stronger the H-A bond. When going down a row in the Periodic Table (see figure below), the atoms get larger so the strength of the bonds get weaker, which means the acids get stronger.
What are factors affecting the strength of acids?
The strength of an acid varies from solvent to solvent. An acid which is strong in water may be weak in a less basic solvent, and an acid which is weak in water may be strong in a more basic solvent. According to Brønsted–Lowry acid–base theory, the solvent S can accept a proton. , which is more acidic than water.
What is the strength of base?
Base strength of a species is its ability to accept H+ from another species (see, Brønsted-Lowry theory). The greater the ability of a species to accept a H+ from another species, the greater its base strength.
Which Oxoacid is strongest?
perchloric acid
The strongest acid is perchloric acid on the left, and the weakest is hypochlorous acid on the far right. Notice that the only difference between these acids is the number of oxygens bonded to chlorine.
What is the strongest superacid?
Fluoroantimonic acid
Fluoroantimonic acid is the strongest superacid based on the measured value of its Hammett acidity function (H0), which has been determined for different ratios of HF:SbF5.
What are 6 strong bases?
Strong Arrhenius Bases
- Potassium hydroxide (KOH)
- Sodium hydroxide (NaOH)
- Barium hydroxide (Ba(OH)2)
- Caesium hydroxide (CsOH)
- Sodium hydroxide (NaOH)
- Strontium hydroxide (Sr(OH)2)
- Calcium hydroxide (Ca(OH)2)
- Lithium hydroxide (LiOH)
How to determine the strength of an acid?
You should commit the strong acids to memory: 1 HCl: hydrochloric acid 2 HNO 3: nitric acid 3 H 2 SO 4: sulfuric acid 4 HBr: hydrobromic acid 5 HI: hydroiodic acid 6 HClO 4: perchloric acid
How is the strength of a covalent bond measured?
The strength of a covalent bond is measured by its bond dissociation energy, that is, the amount of energy required to break that particular bond in a mole of molecules. Multiple bonds are stronger than single bonds between the same atoms.
How to determine the strength of a base?
Strong bases dissociate 100 percent into the cation and OH – (hydroxide ion). The hydroxides of the Group I and Group II metals usually are considered to be strong bases. * These bases completely dissociate in solutions of 0.01 M or less. The other bases make solutions of 1.0 M and are 100 percent dissociated at that concentration.
How does the strength of an acid affect its deprotonation?
The stronger an acid is, the more easily it loses a proton, H +. Two key factors that contribute to the ease of deprotonation are the polarity of the H—A bond and the size of atom A, which determine the strength of the H—A bond. Acid strengths also depend on the stability of the conjugate base.