What did Rutherford expect to happen when the alpha particles hit the foil?
He knew that the -particle had a considerable mass and moved quite rapidly. He therefore anticipated that virtually all of the -particles would be able to penetrate the metal foil, although they would be scattered slightly by collisions with the atoms through which they passed.
When alpha particles were shot at gold foil what happened to the alpha particles quizlet?
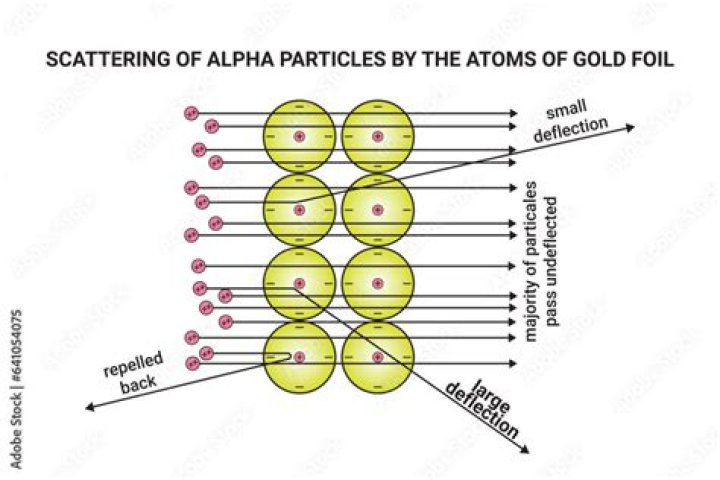
Nearly all alpha particles went straight through the gold foil. (Most of an atom is empty space with a very small nucleus). 1 in 8000 alpha particles were deflected through a wide range of angles. (The nucleus pushes the alpha particles away with an electric force.
Can alpha particles pass through aluminum foil?
The penetrating power of alpha rays, beta rays, and gamma rays varies greatly. Alpha particles can be blocked by a few pieces of paper. Beta particles pass through paper but are stopped by aluminum foil. Gamma rays are the most difficult to stop and require concrete, lead, or other heavy shielding to block them.
Did most alpha particles pass through the gold foil or bounce back backwards?
Since the gold foil was very thin, it was thought that the alpha particles could pass straight through it, or possibly puncture the foil. The scientists were very surprised when other things happened: most of the alpha particles did pass straight through the foil.
Did most alpha particles pass through the gold foil or bounce back backwards quizlet?
Most of the alpha particles went through the foil unaffected. But some of them went off in angles that were not expected. 6. Rutherford proposed that the parts where the alpha particles reflected off, were positive because like charges repel (+/+) .
What happens to the alpha particles as they hit the gold?
Verified by Expert. As the alpha particles hit the gold foil, most of the alpha particles or about 99% passed through the gold foil while those who did not pass through were deflected by small angles and some were rebounded off the gold foil. This experiment was made by Ernest Rutherford and is known as the Gold Foil experiment.
What is the description of the gold foil experiment?
What is the Gold Foil Experiment. Description. The scientists bombarded a thin gold foil of thickness approximately 8.6 x 10-6 cm with a beam of alpha particles in vacuum. They used gold since it is highly malleable, producing sheets that can be only a few atoms thick, thereby ensuring smooth passage of the alpha particles.
Why did Rutherford fire alpha particles at gold foil?
Rutherford fired tiny alpha particles at solid objects such as gold foil. The nucleus was so dense that the alpha particles would bounce off of it, but the electrons were so tiny, and spread out at such great distances, that the alpha particles would pass right through this area of the atom.
What happens if an alpha particle steals an electron?
An alpha would have to “steal” an electron from another atom. This means it would have not only to pull it out of the atom (ionise it, which it does) but accelerate it to the velocity equal its own -velocity. But a fast-going alpha is too fast for that, it will be gone before the electron had enough time to accelerate.