What does corrective risk mean?
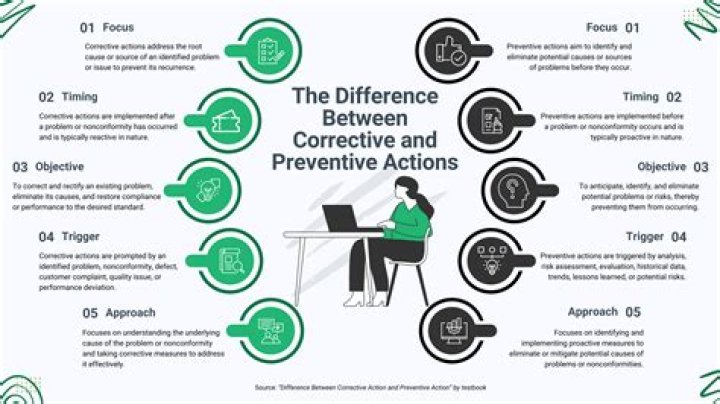
Corrective action: Action taken to eliminate the causes of non-conformities or other undesirable situations, so as to prevent recurrence. Preventive action: Action taken to prevent the occurrence of such non-conformities, generally as a result of a risk analysis.
What is corrective and preventive action procedure?
Corrective Action: Elimination of the cause or causes of an existing nonconformity or undesirable situation in order to prevent recurrence. Preventive Action: Identification and elimination of the cause(s) of potential nonconformities in order to prevent occurrence.
What is the purpose of CAPA?
The purpose of the corrective and preventive action subsystem is to collect information, analyze information, identify and investigate product and quality problems, and take appropriate and effective corrective and/or preventive action to prevent their recurrence.
What is a corrective action example?
For example, putting out a fire in the office is a correction. This action eliminates the problem. Corrective actions, on the other hand, eliminate the root cause of the problem, preventing future issues. The corresponding corrective actions, then, address the root cause of the fire, such as fixing old wiring.
What are corrective measures?
A corrective measure is a measure to resolve existing non-conformities and to prevent their recurrence.
What is the difference between corrective and preventive action?
Corrective action: Action to eliminate the cause of a nonconformity and to prevent recurrence. Preventive action: Action to eliminate the cause of a potential nonconformity or other potential undesirable situation.
What is an example of corrective action?
For example, putting out a fire in the office is a correction. The corresponding corrective actions, then, address the root cause of the fire, such as fixing old wiring. Think of corrective actions as improvements to your organization that you didn’t know you needed until an incident occurs.
What is CAPA example?
Correction and prevention of unacceptable quality system practices should result in fewer nonconformities related to product. For example, it [CAPA] should identify and correct improper personnel training, the failure to follow procedures, and inadequate procedures, among other things.”
How are corrective and preventive actions used to measure?
Paraphrasing from the standard, this requires: 1) Review of actual and potential problems, 2) determining the root cause of the problem (s), 3) Evaluating the need for action, 4) Determining, implementing and documenting (in P&Ps) the action required, and 5) Reviewing the effectiveness of the actions by using data.
How are corrective and preventive actions used in ISO 9001?
This is the third in a series of quick lessons in the 6 required documented procedures of ISO 9001 – 1) Document Control, 2) Record Control, 3) Internal Audit, 4) Control of Non-conforming Product, 5) Corrective Action, and 6) Preventive Action.
Why are preventive / corrective action ( CAPA ) guidelines important?
This is the reason for the implementation of a formal Corrective Action / Preventive Action (CAPA) program. CAPA is a major area of concern for both FDA, ISO 9000, as well as the Automotive and Aerospace industries. They have recognized that how a quality system is maintained and monitored is critical to its effectiveness.
What’s the difference between Corrective and corrective action?
In short, preventive action prevents occurrence and corrective action prevents recurrence, while correction basically refers to containment. If these terms still seem fuzzy, consider the difference between putting out a fire versus eliminating the hazards likely to cause one.