What element has properties like gold?
All Group 11 elements (copper, silver and gold) are relatively inert, corrosion-resistant metals. Copper and gold are colored. These elements have low electrical resistivity so they are used for wiring. Copper is the cheapest and most widely used.
What chemical property could be used to tell the difference between gold and silver?
Hardness helps determine how an element (especially a metal) might be used. Many elements are fairly soft (silver and gold, for example) while others (such as titanium, tungsten, and chromium) are much harder. Carbon is an interesting example of hardness.
What are the 3 chemical properties of gold?
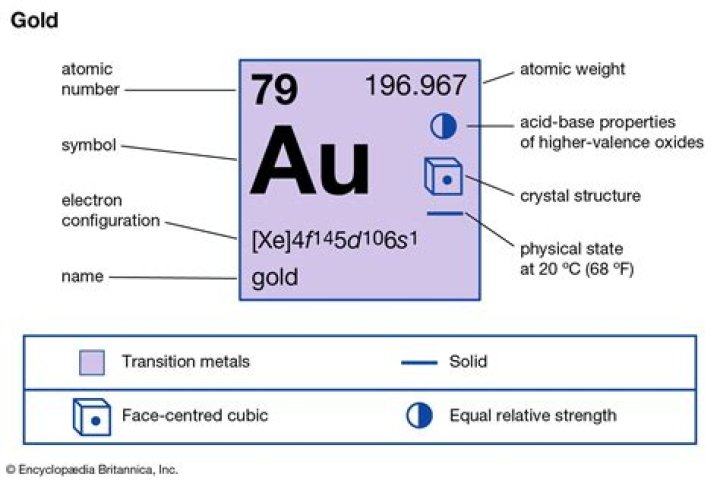
Gold Properties
- Density : 19,300 kg per cubic metre.
- Melting point : 1337.33 K (1064.18 °C).
- Boiling point : 3129 K (2856 °C).
- Atomic number : 79.
- Nucleic protons : 79.
- Atomic weight : 197.
- Nucleic neutrons (normal) : 118.
What are the 5 chemical properties of gold?
What are silvers properties?
Pure silver is nearly white, lustrous, soft, very ductile, malleable, it is an excellent conductor of heat and electricity. It does not oxidize in air but reacts with the hydrogen sulfide present in the air, forming silver sulfide (tarnish). This is why silver objects need regular cleaning. Silver is stable in water.
What are the physical and chemical properties of gold?
Before going through the chemical properties, let us have a look at the physical properties of gold. Gold is ductile as well as malleable in nature. This means it can be drawn into wires and beaten into thin sheets. This soft metal is able to reflect heat as well as light. Gold is a metal and therefore a good conductor of electricity.
Why is gold not a highly reactive compound?
So, generally speaking gold is not a highly reactive compound. Apart from acids, gold does not combine with oxygen, halogens, etc. easily. Aurous (univalent) and auric (trivalent) are two types of compounds formed by gold. Gold is a very good conductor of heat and electricity.
Which is the best description of a chemical property?
1 Chemical properties are properties that can be measured or observed only when matter undergoes a change to become an entirely different kind of matter. 2 Reactivity is the ability of matter to react chemically with other substances. 3 Flammability is the ability of matter to burn.
Why is gold considered to be a noble metal?
As we go into the details of chemical properties of gold, you will understand the connection between the general and physical properties discussed above. Gold is considered to be a noble metal. It is chemically inactive. This is because it is highly resistant to react with other elements.