What experiment disproved the plum pudding model?
gold foil experiment
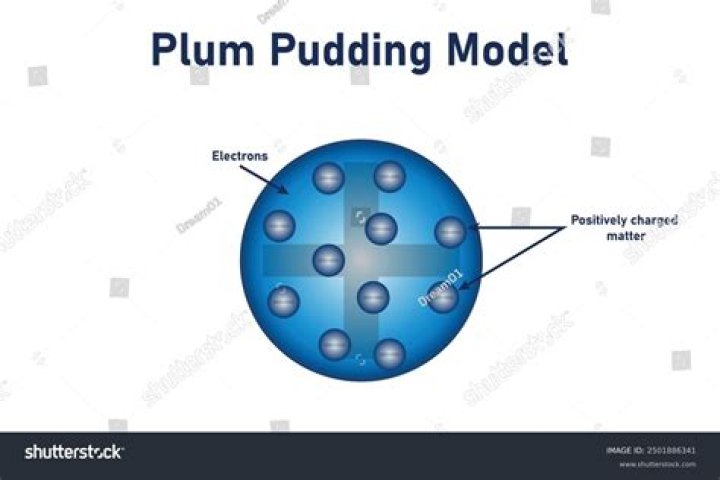
In Thomson’s model, the atom is composed of electrons surrounded by a soup of positive charge to balance the electrons’ negative charges, like negatively charged “plums” surrounded by positively charged “pudding”. The 1904 Thomson model was disproved by Hans Geiger’s and Ernest Marsden’s 1909 gold foil experiment.
How was the plum pudding model proved wrong?
Ernest Rutherford discovered the atomic nucleus using a cathode ray tube. When alpha particles are fired at a thin gold foil, they never go through. Ernest Rutherford proved that the plum-pudding model was incorrect. Ernest Rutherford experimented by firing cathode rays at gold foil.
What model did Rutherford’s experiment disprove?
Thomson’s model of the atom is also known as the plum pudding model. This model describes an atom where the negative charges are surrounded by positive charges like negative plums surrounded by a positively charged pudding. This was disproved by the Gold-foil experiment by Rutherford.
How did these results refute the plum pudding model of the atom?
Rutherfords gold foil experiment involved sending positively charged alpha particles through a thin sheet of gold foil and defecting if there was any deflection of the particles. This result contradicts the plum-pudding model of the atom because the plum pudding model does not explain the deflection of alpha particles.
What is Plum pudding theory?
The ‘plum pudding’ model of the atom was proposed by JJ Thomson, who had also discovered the electron. According to this model, the atom is a sphere of positive charge, and negatively charged electrons are embedded in it to balance the total positive charge. The electrons are like plums in a pudding.
What did the gold foil experiment prove?
The gold-foil experiment showed that the atom consists of a small, massive, positively charged nucleus with the negatively charged electrons being at a great distance from the centre. Niels Bohr built upon Rutherford’s model to make his own.
Why was the plum pudding model rejected?
According to Thomson’s model of the atom, electrons were embedded in a sphere of positive charge and negative and positive charges are equal in magnitude. Hence, Thomson’s model of an atom was rejected.
Why was Rutherford’s model wrong?
The main problem with Rutherford’s model was that he couldn’t explain why negatively charged electrons remain in orbit when they should instantly fall into the positively charged nucleus. This problem would be solved by Niels Bohr in 1913 (discussed in Chapter 10).
Why was the gold foil thin?
Gold was used because it was the only metal that could be rolled out to be very, very thin without cracking. Since the gold foil was very thin, it was thought that the alpha particles could pass straight through it, or possibly puncture the foil. most of the alpha particles did pass straight through the foil.
How did Rutherfords gold foil experiment disprove the plum pudding model?
How did Rutherford’s gold foil experiment disprove the plum pudding model? Chemistry A Closer Look at The Atom Rutherford’s Gold Foil Experiment.
What kind of experiment was the gold foil experiment?
The Experiment. Rutherford’s experiment(9) Rutherford, Geiger, and Marsden shot alpha particles (positively charged particles) at a gold foil surrounded by fluorescent screen. According to Thomson’s plum pudding model, positive and negative charges were distributed evenly within the atom.
Who was the inventor of the plum pudding model?
The plum pudding model was created in 1904 by J.J. Thomson, a physicits who discovered the electron with a cathode ray tube in 1897. Rutherford, Geiger, and Marsden shot alpha particles (positively charged particles) at a gold foil surrounded by fluorescent screen.
How are alpha particles deflected in plum pudding model?
Well, there is scattering AND scattering. True, both the plum pudding model and the “planetary” model predict that alpha particles should be deflected, but the deflections predicted by the two models are very different. In the plum pudding model, one assumes that the charge of the protons is distributed and fills all the atom volume.