What happened to the alpha particles as they hit the gold?
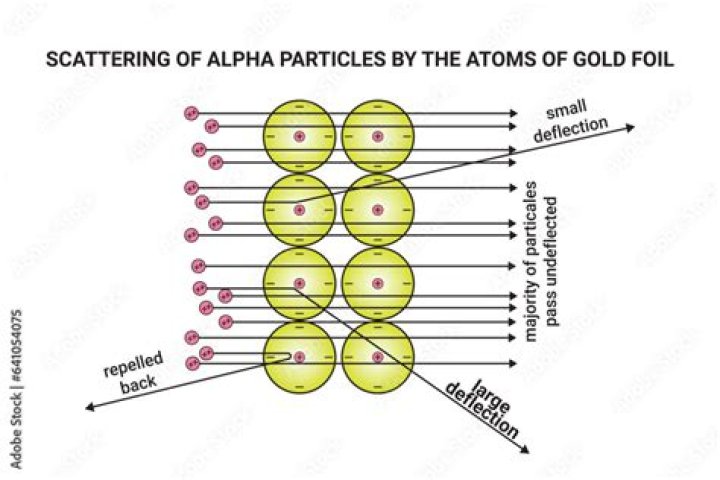
A: The alpha particles would penetrate the gold foil. Alpha particles are positive, so they might be repelled by any areas of positive charge inside the gold atoms. Most of the alpha particles passed straight through the gold foil as though it wasn’t there. The particles seemed to be passing through empty space.
Why an α particle and a gold nucleus repel each other?
Explanation: Alpha particles are are positively charges particles that are made up of 2 protons, 2 neutrons and zero electrons. Due to the fact that protons have a +1 charge and neutrons hold no charge, this would give the particle a +2 charge over all. This is due to the fact that like charges repel each other.
How does the gold foil experiment work?
Most alpha particles passed straight through the gold foil, which implied that atoms are mostly composed of open space. Some alpha particles were deflected slightly, suggesting interactions with other positively charged particles within the atom.
What is the significance of the gold foil experiment?
The Geiger–Marsden experiments (also called the Rutherford gold foil experiment) were a landmark series of experiments by which scientists learned that every atom has a nucleus where all of its positive charge and most of its mass is concentrated.
Why are particles deflected bounced backwards?
A tiny number of alpha particles, traveling at 10% of the speed of light, hit a dense atomic center right in its middle. The collision and the repulsion cause the alpha particle to “bounce” backwards and move on a very different path. These are the reflected rays.
Why do most alpha particles pass through gold foil?
a small number of alpha particles were deflected by large angles (> 4°) as they passed through the foil….Rutherford and the nucleus.
| What happened | Rutherford’s conclusions |
|---|---|
| Most of the alpha particles did pass straight through the foil. | The atom being mostly empty space. |
What did the alpha scattering experiment reveal?
Rutherford’s gold foil experiment demonstrated that almost all of the mass of an atom is in a tiny volume in the center of the atom which Rutherford called the nucleus. This positively charged mass was responsible for deflecting alpha particles propelled through the gold foil.
What happens when an alpha particle collides with a gold nucleus?
when the alpha particle collides with the gold nucleus, there are 2 quantities that are conserved: linear momentum and energy. The kinetic energy lost by the alpha particle will be used to overcome the electric potential energy between the particles and the kinetic energy of the gold nucleus.
What is the initial momentum of the alpha particle?
The particles are initially far apart, and the gold nucleus is initially at rest. Answer the following questions about the collision. What is the initial momentum of the alpha particle?
What’s the Ke of the alpha particle at its closest approach?
Assuming that the gold nucleus is essentially stationary (as proposed in the problem statement), what’s the KE of the alpha particle when it’s at its closest approach? Where’s all the energy? Right.
When does an alpha particle bounce off an atom?
When the electric potential energy of an atom is larger than the kinetic energy of an oncoming alpha particle, the particle simply bounces off it. I tried out a practice problem with slightly different values and the alpha particle’s final momentum was negative (not 0), and only slightly smaller a number than its initial momentum.