What happens to an atom when you add or subtract a proton?
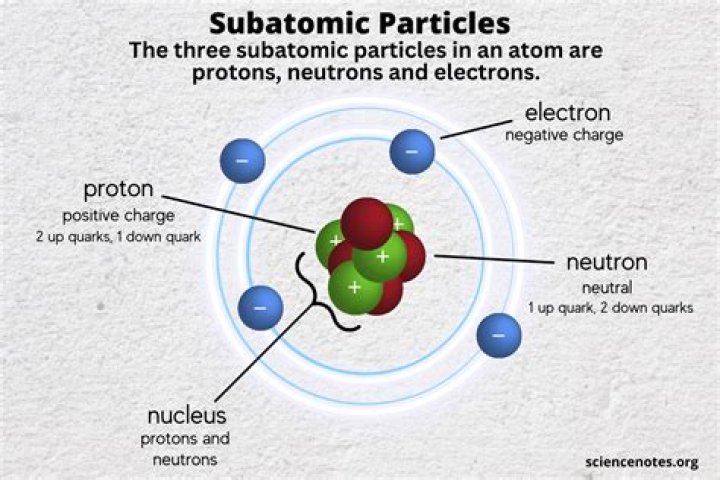
If you add or subtract a proton from the nucleus, you create a new element. If you add or subtract a neutron from the nucleus, you create a new isotope of the same element you started with. In a neutral atom, the number of positively charged protons in the nucleus is equal to the number of orbiting electrons.
Do protons make the atom more positive or negative?
A neutron has no charge, whereas a proton has a positive charge that exactly balances the negative charge on an electron. Table 1 lists the charges of these three fundamental particles, and gives their masses expressed in atomic mass units….Atomic Structure.
| Particle | Charge | Mass (amu) |
|---|---|---|
| Protons | +1 | 1.00782 |
| Neutrons | 0 | 1.00867 |
Which particle in the atom has the highest density?
Where is the most dense area of the atom? The nucleus is the small, positively-charged center of the atom, which is comprised of protons and neutrons. The nucleus is relatively high mass and small volume, therefore it is a very dense region of the atom.
What happens when atoms have more protons?
If it has a net charge, (more or less electrons than protons) it is an ion. If it has more electrons than protons it has a net negative charge and is known as an anion. If it has fewer electrons than protons it has a net positive charge and is known as a cation.
What would happen if there were no neutrons?
Simply, if there are no neutrons, the protons would get separated from the atom by repelling each other and thus the nuclei will be left alone. Therefore there will be no further elements.
Which particle if lost from the nucleus will not result in a change in the atomic mass number?
Therefore, when a nucleus loses a neutron the atomic number does not change.
Why is 99 empty space?
Atoms make up everything, but they also exist very, very far apart – and atoms themselves are more void than they are matter. Every atom has a nucleus surrounded by electrons. Every human on planet Earth is made up of millions and millions of atoms which all are 99% empty space.
How much of atom is empty space?
99.9999999%
But it might humble you to know that all of those things — your friends, your office, your really big car, you yourself, and everything in this incredible, vast universe — are almost entirely, 99.9999999%, empty space.