What happens when you put gold in acid?
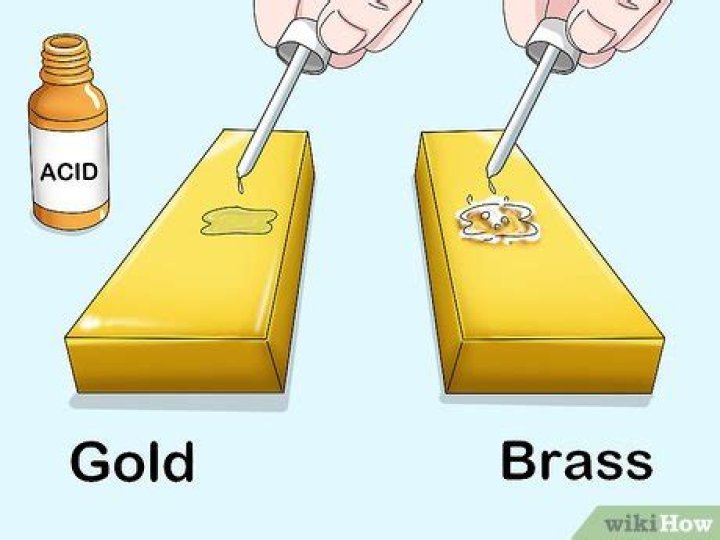
Muriatic acid, better known today as hydrochloric acid, is a simple, corrosive liquid with well-studied chemical properties. When gold is subjected to treatment with muriatic acid alone, nothing happens. But when \muriatic acid is combined with nitric acid to treat gold, the gold dissolves.
Does acid destroy gold?
While hydrochloric acid, by itself, will not react with or harm gold in any way, gold and other precious, non-reactive metals can be dissolved by a mixture of hydrochloric acid and nitric acid, in a three-to-one ratio by volume.
What metals will dissolve in sulfuric acid?
Sulphuric acid is an oxidizing acid and can dissolve also noble metals such as copper. As a strong dibasic acid H2SO4 dissolves all base metals by forming hydrogen and the corresponding sulfates and hydrogen sulfates.
What metal does not dissolve in sulfuric acid?
Elements below hydrogen in the electrochemical series don’t react with sulphuric acid. They include Copper, Silver, Gold etc. Elements like Platinum and Gold are called noble metals too as they don’t react with most of the compounds or other reactants.
What would happen if a gold ring is left in a weak acid?
Answer: The acid test for gold is to rub the gold-colored item on black stone, which will leave an easily visible mark. If the mark remains, it is tested by applying aqua regia (nitric acid and hydrochloric acid). If the mark is removed, then this test dissolves the gold, proving the item to be genuine gold.
What is the easiest way to test gold?
One of the simplest preliminary tests for gold at home is the Float Test. All you need is a cup (or bowl) of water and your gold item. All you have to do is place your gold piece into the water! If it’s genuine gold, then it will immediately sink to the bottom of the cup.
What acid will not dissolve gold?
Now the exciting thing about Aqua Regia is that neither of the two acids by themselves can dissolve gold. Nitric acid doesn’t really react with gold very much at all, and HCl, hydrogen chloric acid can’t get a hold of the gold to get the reaction going.
What is the strongest acid on earth?
Fluoroantimonic acid
Fluoroantimonic acid is the strongest super-acid known in existence. It is 20 quintillion times more acidic than 100% sulfuric acid, and it can dissolve glass plus a host of other substances.
What can you not mix with sulfuric acid?
Some chemicals are NOT to be mixed!
| DON’T MIX | WITH |
|---|---|
| Sulfur dichloride (SCl2) | Water, alcohols. |
| Sulfuric acid (H2SO4) or oleum | Chlorates, perchlorates, permanganates, water. |
| Sulfur tetrachloride (SCl4) | Water. |
| Silver (Ag) and its salts | Acetylene, hydrogen, oxalic acid, tartaric acid, ammonia, ammonium salts. |
How fast does sulfuric acid gas kill?
The deadly numbers and statistics from H2S gas 800 ppm is the lethal concentration for five minutes of exposure. Concentrations over 1000 ppm cause immediate collapse with loss of breathing, even after inhalation of a single breath – death can occur within one to four hours of exposure.
Is it possible to dissolve gold in sulfuric acid?
The answer is yes, and no. According to a 1940 patent #2185858, gold or palladium are stripped from a copper based substrate by making the parts anodic in a fairly concentrated sulfuric acid solution. According to the patent, persulfuric acid is produced at the anode surface, which momentarily dissolves the gold.
What are the effects of sulfuric acid on metal?
Diluted sulfuric acid (H 2 SO 4) and magnesium, for example, will react vigorously: Mg + H 2 SO 4 -> MgSO 4 + H 2. In practice, not all of these metals will react with sulfuric acid under normal circumstances. Although the pure metals will react, some elements, when exposed to air, quickly acquire a layer of oxide.
What happens if you touch sulphuric acid?
For dilute and weak acids, there’d be no effect whatsoever. The sulphuric acid in a car battery will cause a very nasty burn, however. As will muriatic acid (concentrated HCl). Hydrofluoric acid will slowly dissolve your tissues in a very agonizing way.
What can be used as a precipitant for gold?
The most common gold precipitants are some form of sulfite, such as sodium sulfite, sodium bisulfite, or sodium metabisulfite. Less common but still viable gold precipitants are ferrous sulfate or oxalic acid. If the worker uses too much nitric acid in the aqua regia,…