What industry is calorimetry used in?
Calorimeters are mainly used in the coal industry, i.e. coal fired power stations, iron and steel plants, cement plants and other users of coal. However, they are often used in other non-coal related industries.
Why is calorimetry important in industry?
In either case, reaction calorimetry can provide critical process-safety information before scale-up, allowing scientists and engineers to make appropriate decisions and create a robust process that ensures products are processed according to plant capabilities.
What are the uses of calorimeter?
A calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. For example, when an exothermic reaction occurs in solution in a calorimeter, the heat produced by the reaction is absorbed by the solution, which increases its temperature.
How is calorimetry used in the real world?
Calorimetry also plays a large part of everyday life, controlling the metabolic rates in humans and consequently maintaining such functions like body temperature. Constant Volume (bomb) calorimetry, is used to measure the heat of a reaction while holding volume constant and resisting large amounts of pressure.
What are the two basic types of calorimeter?
Lesson Summary Calorimeters are equipment used to measure the enthalpy, which is the heat energy, of a reaction. There are two commonly used types of calorimeters: coffee cup calorimeters and bomb calorimeters.
Why bomb calorimeter is used?
Bomb calorimeters are devices used to determine the heat of combustion of a chemical reaction. The information gathered from a bomb calorimeter during a chemical reaction tells scientists whether certain products are safe for use and the quality level of each product being tested.
What is the most important part of a calorimeter?
A simple calorimeter just consists of a thermometer attached to a metal container full of water suspended above a combustion chamber. According to this, the most important part would be the heat fusion of water, basically because this value will remain the same regardless the food you are using of the metal.
What is the principle of calorimeter?
Calorimeter Principle The body at higher temperature releases heat while the body at lower temperature absorbs heat. The principle of calorimetry indicates the law of conservation energy, i.e. the total heat lost by the hot body is equal to the total heat gained by the cold body.
Why is enthalpy important in real life?
Change in enthalpy can apply to refrigerators and hand warmers. In a fridge, refrigerants such as Freon are evaporated. The enthalpy of vaporization (liquid to gas energy change) is equivalent to the coldness of your food. Some people use chemical heat packs (hand warmers) outside.
What is calorimeter explain?
A calorimeter is an object used for calorimetry, or the process of measuring the heat of chemical reactions or physical changes as well as heat capacity. Multiplying the temperature change by the mass and specific heat capacities of the substances gives a value for the energy given off or absorbed during the reaction.
How are calorimeters used in the chemical industry?
A calorimeter is a device used for calorimetry, or measuring heat capacity or the heat of physical changes or chemical reactions. In pharmaceuticals, they are used in drug design. In the chemical industry, they are used for quality control, and in biological studies, they are used for metabolic rate examination.
How is the cal3k calorimeter used in the coal industry?
How the CAL3K Calorimeter can be used in the coal industry. The coal industry is the traditional application of calorimeters because coal has a variety of properties, apart from being black. If the coal is used for steam generation then the calorific value is of paramount importance.
What is the purpose of a bomb calorimeter?
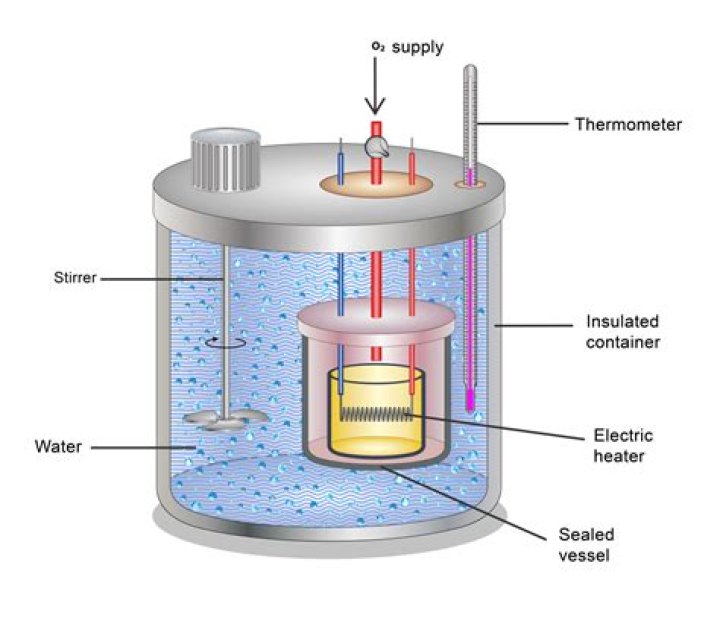
Bomb Calorimeter: Also known as a constant-volume calorimeter, this is built to withstand the pressure that builds up as a result of a reaction as air heats in the reaction vessel. The change in water temperature is used to calculate the heat of combustion.
Why is a thermometer used in a calorimeter experiment?
The vessel is filled with water and the fuel is burned leading to the heating of water. Heat loss by the fuel is equal to the heat gained by the water. This is why it is important to insulate the calorimeter from the environment; to improve the accuracy of the experiment. This change in heat can be measured through the thermometer.