What is a Bohr model of gold?
Rutherford’s gold foil experiment demonstrated that almost all of the mass of an atom is in a tiny volume in the center of the atom which Rutherford called the nucleus. This positively charged mass was responsible for deflecting alpha particles propelled through the gold foil.
What is the atomic makeup of gold?
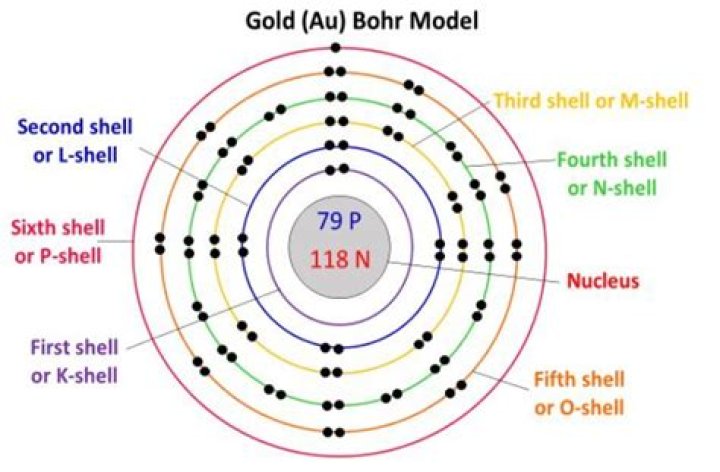
79 protons
Gold is the chemical element with 79 protons in each atomic nucleus. Every atom containing 79 protons is a gold atom, and all gold atoms behave the same chemically.
What is gold valence electrons?
How Many Valence Electrons are in Gold. A number of valence electrons are a number of electrons which are present at the outer most shell of the atom. Gold has 79 electrons and has only one electron in the outermost shell and it makes univalent compounds. So, the gold has only 1 (one) valence electron.
What structure is gold?
| Gold | |
|---|---|
| Spectral lines of gold | |
| Other properties | |
| Natural occurrence | primordial |
| Crystal structure | face-centered cubic (fcc) |
Who disproved Bohr’s model?
Five years later, the model would be disproved by Hans Geiger and Ernest Marsden, who conducted a series of experiments using alpha particles and gold foil – aka. the “gold foil experiment.”
How can we find Valency of gold?
Complete answer: As we can see that the valence shell of the gold atom is 6 and there is 1electron in this valence shell, so it can lose this electron to form a +1 oxidation state, therefore, having a valency 1.
When did Niels Bohr come up with the Bohr model?
Here’s a closer look at the Bohr Model, which is sometimes called the Rutherford-Bohr Model. Niels Bohr proposed the Bohr Model of the Atom in 1915.
Which is an example of the Bohr model?
The simplest example of the Bohr Model is for the hydrogen atom (Z = 1) or for a hydrogen-like ion (Z > 1), in which a negatively-charged electron orbits a small positively-charged nucleus. Electromagnetic energy will be absorbed or emitted if an electron moves from one orbit to another.
Why is the Bohr model of the atom called the Rutherford model?
Overview of the Bohr Model Niels Bohrproposed the Bohr Model of the Atom in 1915. Because the Bohr Model is a modification of the earlier Rutherford Model, some people call Bohr’s Model the Rutherford-Bohr Model. The modern model of the atom is based on quantum mechanics.
When did Erwin Schrodinger change the Bohr model?
Ultimately, the Bohr model and models based upon it were replaced Wolfgang Pauli’s model based on quantum mechanics in 1925. That model was improved to produce the modern model, introduced by Erwin Schrodinger in 1926.