What is aqueous silver nitrate?
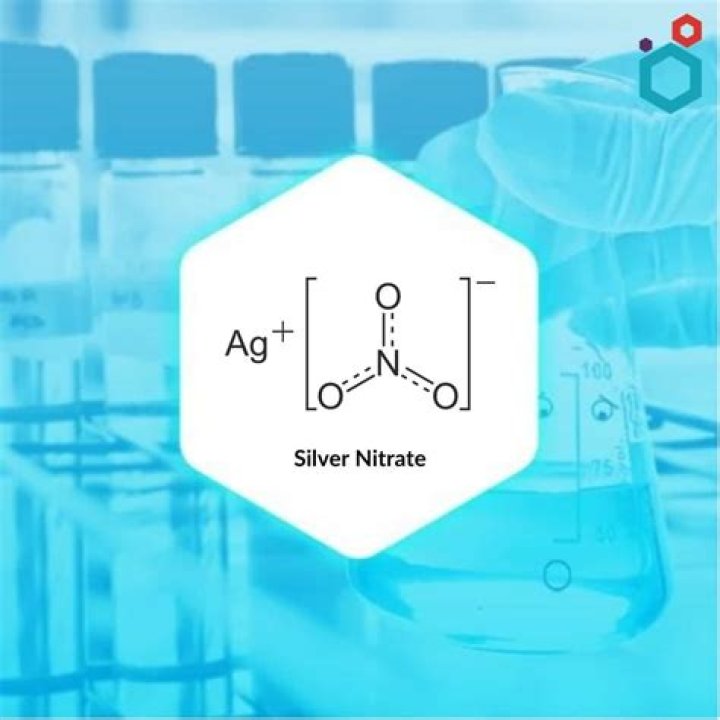
In analytical chemistry, aqueous solutions of silver nitrate are used in the volumetric determination of halides, cyanides, and thiocyanates, as well as for the detection of reducing agents and of the cations of various acids that form insoluble silver salts. At a red heat, silver is formed.
Is AgNO3 aqueous or liquid?
AgNO3 as a colourless appearance in its solid state and is odourless. In its solid state, it has a density of 4.35 grams per cubic centimetre. Its density in the liquid state at a temperature of 210oC corresponds to 3.97 g/cm3. The melting and boiling points of silver nitrate are 482.8 K and 713 K respectively.
Can AgNO3 be aqueous?
For example, when an aqueous solution of silver nitrate (AgNO3) is added to the aqueous solution of sodium chloride (NaCl), a white precipitate of silver chloride (AgCl) is formed that is indicated by the following chemical reaction. Ionic compounds dissociate into ions when dissolved in water (aqueous solution).
Does silver nitrate dissolve in water?
Water
Glycerol
Silver nitrate/Soluble in
What would you observe if A is added to a solution of silver nitrate?
When a few drops of a silver nitrate solution are added to a slightly acidic aqueous solution that contains chloride ions, a white precipitate of silver chloride will form.
What is the chemical equation for silver nitrate?
This reaction is commonly used to illustrate basic solubility rules, and solubility equilibria. All nitrates are soluble, hence silver nitrate is soluble; and all halides are soluble, EXCEPT for AgX, PbX_2, and Hg_2X_2. Thus silver nitrate is soluble, but silver chloride precipitates from solution as a curdy white solid.
Which is soluble silver nitrate or silver iodide?
Thus silver nitrate is soluble, but silver chloride precipitates from solution as a curdy white solid. We write the net ionic equation as: Silver chloride is a curdy white solid; silver bromide is a creamy yellow; and silver iodide is bright yellow.
What can be used to make silver nitrate?
Silver nitrate can be prepared by reacting silver, such as a silver bullion or silver foil, with nitric acid, resulting in silver nitrate, water, and oxides of nitrogen. Reaction byproducts depend upon the concentration of nitric acid used.
What happens when silver nitrate is heated to ethanol?
The reaction between silver nitrate and ethanol is explosive. The silver present in this compound is displaced by copper, which forms copper nitrate. The chemical equation for this reaction is given by 2AgNO 3 + Cu → Cu(NO 3) 2 + 2Ag; When heated to 440 o C, this compound completely decomposes to give oxygen, nitrogen dioxide, and silver.