What is meant by the strength or concentration of an acid?
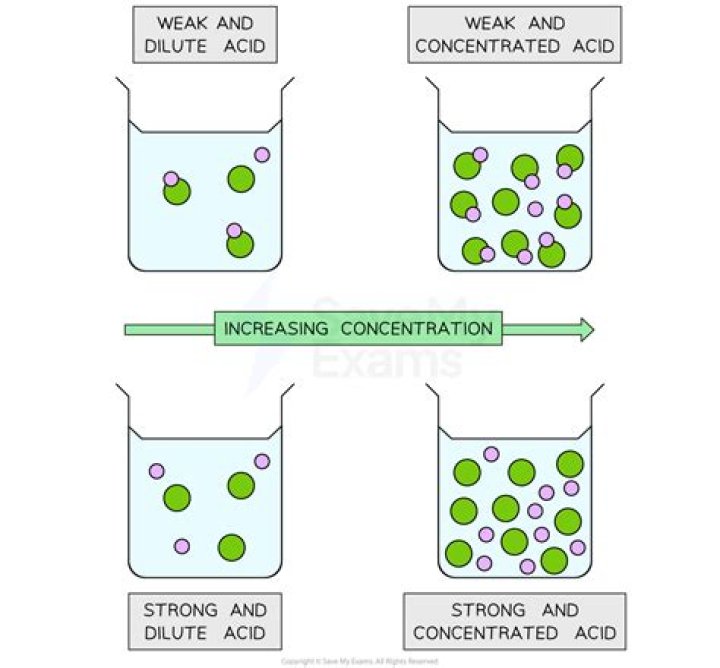
The strength of an acid relates to the number of free ions in solution while the concentration of an acid relates to the number of ions that it contributes to a solution.
What is a weak acid Example?
Weak acids are that acid that can be ionized partially in their solutions, whereas strong acids are completely ionized when dissolved in water. A weak acid is more commonly used compared to strong acids. They are found in daily life like vinegar (acetic acid) and lemon juice (citric acid).
Which is the strongest acid in the following?
Since, in HClO4, oxidation number of Cl is highest, so, HClO4 is the strongest acid among the given acids.
Is hydrochloric acid a weak or strong acid?
HCl is a strong acid because it dissociates almost completely. By contrast, a weak acid like acetic acid (CH3COOH) does not dissociate well in water – many H+ ions remain bound-up within the molecule.
What are 2 weak acids?
Some common examples of weak acids are listed below.
- Formic acid (chemical formula: HCOOH)
- Acetic acid (chemical formula: CH3COOH)
- Benzoic acid (chemical formula: C6H5COOH)
- Oxalic acid (chemical formula: C2H2O4)
- Hydrofluoric acid (chemical formula: HF)
- Nitrous acid (chemical formula: HNO2)
Who is the weakest acid?
Hydrofluoric acid is the only weak acid produced by a reaction between hydrogen and halogen (HF). Acetic acid (CH3COOH), which is contained in vinegar, and oxalic acid (H2C2O4), which is present in some vegetables, are examples of weak acids.
Which is the best definition of acid strength?
Acid Strength Definition Acid strength can be defined as the tendency of an acid, to dissociate into a proton, H+, and an anion, A−, and symbolized by the formula HA. The dissociation of a robust acid in solution is effectively complete, except in its most concentrated solutions HA → H+ + A−
How many strong acids are there in the world?
There are six strong acids. The others are considered to be weak acids. You should commit the strong acids to memory: If the acid is 100 percent dissociated in solutions of 1.0 M or less, it is called strong.
How is the concentration of a strong acid equal to its concentration?
This generally means that in aqueous solution at standard temperature and pressure, the concentration of hydronium ions is equal to the concentration of strong acid introduced to the solution. Ionization of acids and bases in water: A strong acid ionizes completely in an aqueous solution by losing one proton (H+).
Which is a better measure of acidity, pH or value?
For concentrated solutions of acids, especially strong acids for which pH < 0, the value is a better measure of acidity than the pH. Image of a strong acid mostly dissociating.