What is the chemical name for silver nitrate?
AgNO3
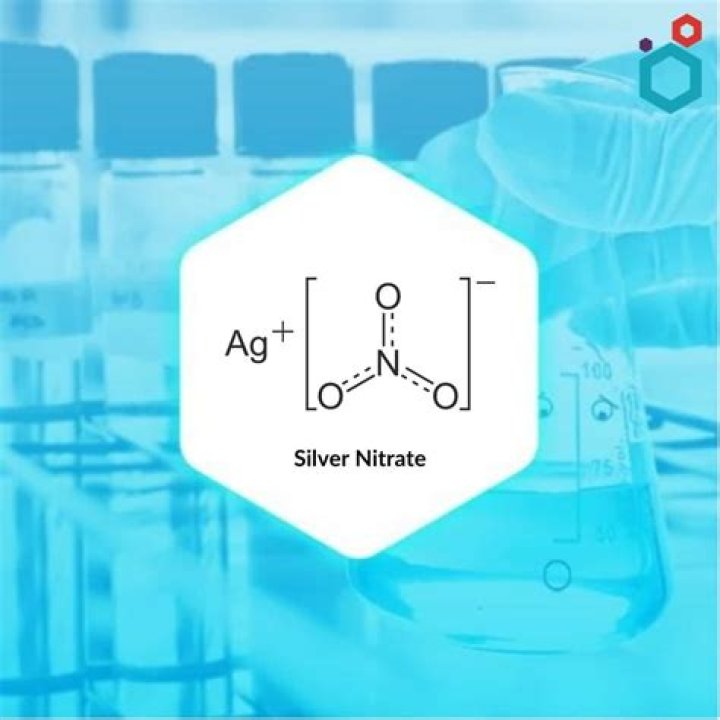
Silver nitrate is an inorganic compound with chemical formula AgNO3.
What compound is nano3?
Sodium nitrate
Sodium nitrate/IUPAC ID
What are the hazards of silver nitrate?
► Exposure to Silver Nitrate can cause headache, dizziness, nausea and vomiting. to transport Oxygen, causing headache, fatigue, dizziness, and a blue color to the skin and lips (methemoglobinemia). Exposure to very high levels can cause trouble breathing, collapse and even death.
Is silver nitrate poisonous?
On average, 3.8 grams of silver nitrate given orally can cause argyria to begin to be observed. Silver nitrate is mainly considered a poison with ingestion due to the corrosive nature of the compound. If ingested, silver nitrate can cause potentially fatal gastroenteritis and gastrointestinal bleed.
Is salt a nitrate?
Sodium nitrate is a kind of salt that has long been used to preserve foods. Sodium nitrate can be found in plants and unregulated drinking water. Nitrogen is turned into sodium nitrate in soil and is necessary for plant growth. Plants absorb sodium nitrate from the soil in varying amounts.
What is the chemical name of sodium hydroxide?
Sodium hydroxide
Sodium oxidanide
Sodium hydroxide/IUPAC ID
Is silver nitrate painful?
Silver nitrate sticks are used to help treat and remove granulation tissue around your child’s stoma. Do not use silver nitrate if your child has a sensitivity to silver. Applying silver nitrate may be painful. Giving your child acetaminophen or ibuprofen before application may help your child stay comfortable.
Which is the correct formula for silver nitrate?
Silver nitrate is an inorganic compound with the chemical formula AgNO3. In its solid form, silver nitrate is coordinated in a trigonal planar arrangement. It is often used as a precursor to other silver -containing compounds.
What can silver nitrate be used for in medicine?
Silver Nitrate is an inorganic chemical with antiseptic activity. Silver nitrate can potentially be used as a cauterizing or sclerosing agent. Silver(1+) nitrate is a silver salt and an inorganic nitrate salt.
What is the reaction between silver nitrate and ethanol?
The hazards of AgNO 3 include its toxic and corrosive nature. The reaction between silver nitrate and ethanol is explosive. The silver present in this compound is displaced by copper, which forms copper nitrate. The chemical equation for this reaction is given by 2AgNO 3 + Cu → Cu(NO 3) 2 + 2Ag.
What happens to silver nitrate at 350 °C?
On exposure to air or light in the presence of organic matter, silver nitrate becomes grey or greyish-black. In the presence of a trace of nitric acid, silver nitrate is stable to 350 °C. Kirk-Othmer Encyclopedia of Chemical Technology. 3rd ed., Volumes 1-26.