What is the compound element of gold?
Gold is a chemical element with symbol Au and atomic number 79….7.1Element Forms.
| CID | 105093 |
|---|---|
| Name | gold(3+) |
| Formula | Au+3 |
| SMILES | [Au+3] |
| Molecular Weight | 196.967 |
Can gold Make compounds?
Although gold is the most noble of the noble metals, it still forms many diverse compounds. The oxidation state of gold in its compounds ranges from −1 to +5, but Au(I) and Au(III) dominate its chemistry.
What is gold compound name?
Gold
| PubChem CID | 23985 |
|---|---|
| Chemical Safety | Laboratory Chemical Safety Summary (LCSS) Datasheet |
| Molecular Formula | Au |
| Synonyms | 7440-57-5 Au Gold Colloidal gold Gold powder More… |
| Molecular Weight | 196.96657 |
Is gold a compound or element?
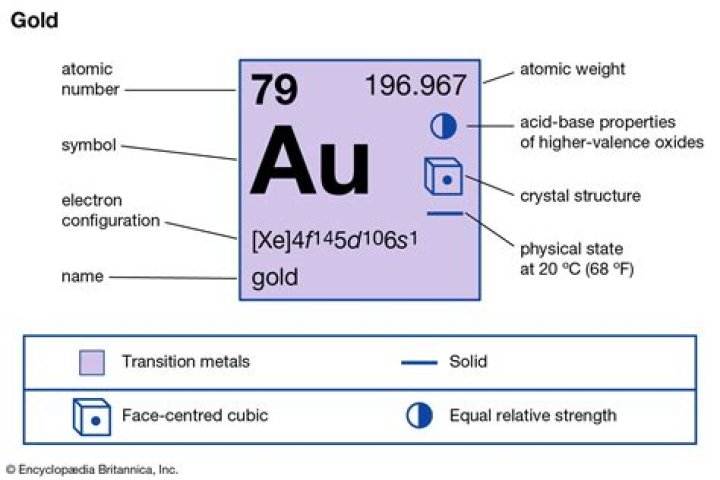
Gold (Au), chemical element, a dense lustrous yellow precious metal of Group 11 (Ib), Period 6, of the periodic table.
What is the unique characteristics of gold?
Gold has several qualities that have made it exceptionally valuable throughout history. It is attractive in colour and brightness, durable to the point of virtual indestructibility, highly malleable, and usually found in nature in a comparatively pure form.
What kind of compounds are found in gold?
Gold: compounds information 1 Hydrides. The term hydride is used to indicate compounds of the type M x H y and not necessarily to indicate that any… 2 Fluorides. 3 Chlorides. 4 Bromides. 5 Iodides. 6 Oxides. 7 Sulfides. 8 Selenides. 9 Tellurides. 10 Nitrides. 11 Carbonyls. 12 Complexes. More …
Why is gold found in the metallic form?
Gold is characterised chemically by an extreme indifference to the action of all bodies usually met with in nature. Its simpler compounds are formed with difficulty, and decompose readily, especially when heated. The result is that gold is found in nature chiefly in the metallic form, and the mineralogist has, therefore, few compounds to consider.
What kind of reaction does gold have with water?
Gold does not react with water. Gold metal reacts with chlorine, Cl 2, or bromine, Br 2, to form the trihalides gold (III) chloride, AuCl 3, or gold (III) bromide, AuBr 3, respectively. On the other hand, gold metal reacts with iodine, I 2, to form the monohalide gold (I) chloride, AuI.
Which is the correct formula for the compound gold?
The gold in them is not present as an elementary ion, but forms part of complex ions. Gold forms two series of simple compounds, having the general formulae AuR and AuR3, while doubtful compounds corresponding to AuR2, AuR4, and AuR4, have been declared to exist by Thomsen, Prat, Figuier, and others.