What is the formula for silver II bromide?
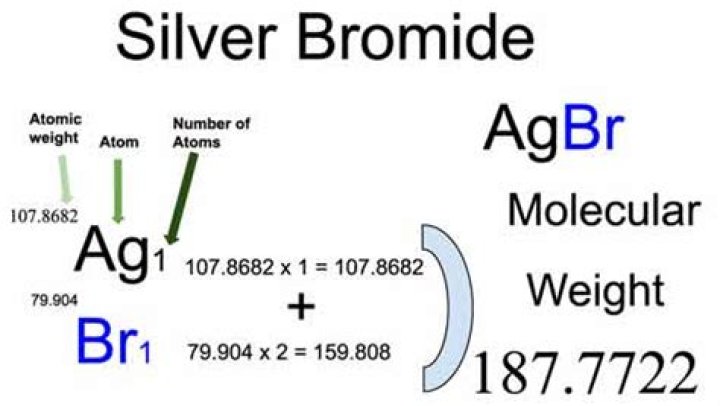
Formula and structure: The chemical formula of silver bromide is AgBr and its molar mass is 187.77 g/mol.
What is the correct formula for Ag+ Br?
Silver;bromide | AgBr – PubChem.
Is br2 an element?
Bromine is a chemical element with the symbol Br and atomic number 35….
| Bromine | |
|---|---|
| Phase at STP | liquid |
| Melting point | (Br2) 265.8 K (−7.2 °C, 19 °F) |
| Boiling point | (Br2) 332.0 K (58.8 °C, 137.8 °F) |
What is the name of br2?
Bromine Dibromine
Bromine
| PubChem CID | 24408 |
|---|---|
| Structure | Find Similar Structures |
| Chemical Safety | Laboratory Chemical Safety Summary (LCSS) Datasheet |
| Molecular Formula | Br2 |
| Synonyms | 7726-95-6 Br Bromine Dibromine Brom More… |
What is the charge for silver bromide?
When the atoms combine to form silver bromide the electron from the outer shell of the silver atom moves over to the bromine and so gives it an overall negative charge (—e). The loss of the electron from the silver makes it have an overall positive charge (+e).
Is silver bromide dangerous?
Harmful by ingestion, inhalation and skin contact. Prolonged exposure may result in argyria, a bluish discolouration of the skin. Irritating to eyes and may irritate skin. Dangerous for the environment.
What is the formula for K and Br?
KBr
Potassium bromide/Formula
What is the formula of silver chloride?
AgCl
Silver chloride/Formula
Can bromine kill you?
Bromine vapors are very toxic with inhalation. Humans can absorb organic bromines through the skin, with food and during breathing. Organic bromines are widely used as sprays to kill insects and other unwanted pests. In many cases they are poisonous to humans, too.
Why is bromine written as Br2?
a) Why is bromine written as Br2 on the right side? Bromine is a diatomic molecule and always needs to be bonded to something; even bonding to itself works. The Br is already bonded to something else (the Li). If it were written LiBr2 it would not be a neutral compound.
How to find the molecular formula for bromide?
Bromide ion PubChem CID 259 Structure Find Similar Structures Molecular Formula Br- Synonyms bromide bromide ion 24959-67-9 UNII-9529 Molecular Weight 79.90
What is the chemical structure of sodium bromide?
Sodium bromide is formed by one sodium cation Na + and one bromide anion Br – which are joined through an ionic bond. The crystal structure has the form of a face-centered cubic with six bromide anions surrounded one sodium cation. Its chemical structure can be written as below, in the common representations used for organic molecules.
Which is the best description of silver bromide?
Silver bromide (AgBr), a soft, pale-yellow, water-insoluble salt well known (along with other silver halides) for its unusual sensitivity to light. This property has allowed silver halides to become the basis of modern photographic materials.
What are the P codes for silver bromide?
H400 (80%): Very toxic to aquatic life [ Warning Hazardous to the aquatic environment, acute hazard] H410 (100%): Very toxic to aquatic life with long lasting effects [ Warning Hazardous to the aquatic environment, long-term hazard] (The corresponding statement to each P-code can be found at the GHS Classification page.)