What is the formula silver sulfate?
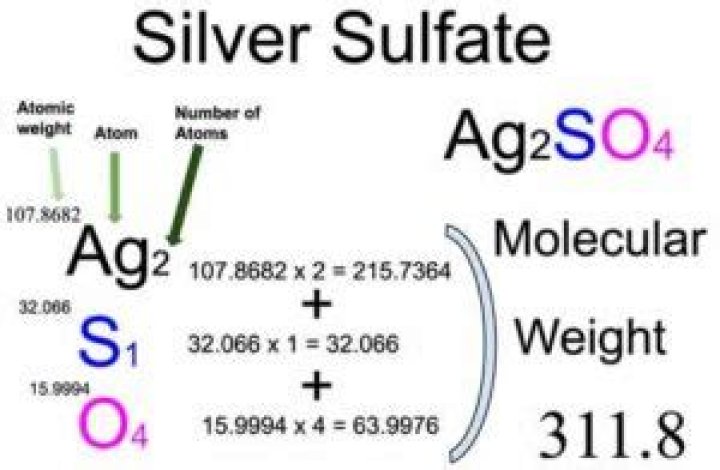
Ag2SO4
Silver sulfate/Formula
What is the name of AgHSO4?
Silver Hydrogen Sulfate AgHSO4 Molecular Weight — EndMemo.
What is the formula for hydrogen and sulfate?
Hydrogen sulfate also commonly known as bisulfate is an anion. The hydrogen sulphate formula is given by HSO4-….Physical Properties of Hydrogen Sulfate.
| Chemical Formula of hydrogen sulfate | HSO4- |
|---|---|
| Reactive With | Water, Nitric Acid, Ethanol, Acetone |
How many ions are in Ag2SO4?
Since silver(I) has a +1 charge, and sulfate has a -2 charge, two silver ions are required to bond with one sulfate ion. This gives us the formula Ag2 SO4.
What is the color of silver sulphate?
Silver sulfate
| PubChem CID | 159865 |
|---|---|
| Molecular Weight | 311.80 |
| Component Compounds | CID 23954 (Silver) CID 1118 (Sulfuric acid) |
| Dates | Modify 2021-07-17 Create 2005-08-08 |
| Description | Silver sulfate is an odorless white to gray solid. Sinks and mixes with water. (USCG, 1999) CAMEO Chemicals |
What is Iodite formula?
Iodite | IO2- – PubChem.
What color is silver sulfate?
white
Silver(I) sulfate is white.
What is the Lewis structure of HSO4?
For the HSO4- Lewis structure, we have a total of 32 valence electrons. That includes this one up here. We’ll put a pair of electrons between atoms to form chemical bonds–we’ve used 8 and 10. Then we’ll go around and fill the octets for the Oxygen; 12, 14, and 32.
Is hydrogen sulfate a strong acid?
This mixture of sulfuric acid and water boils at a constant temperature of 338 °C (640 °F) at one atmosphere pressure. Sulfuric acid is a very strong acid; in aqueous solutions it ionizes completely to form hydronium ions (H3O+) and hydrogen sulfate ions (HSO4−).
What is the chemical formula for hydrogen sulfate?
What is Hydrogen sulfate? Hydrogen Sulfate is an anion with the chemical formula HSO4–. It forms sulfuric acid when exposed to water. HSO 4– very small hydrogen sulfate ion.
What is the chemical formula for silver sulfate?
Silver sulfate (Ag2SO4) is an ionic compound of silver used in silver plating and as a non-staining substitute to silver nitrate. This sulfate is stable under ordinary conditions of use and storage, though it darkens upon exposure to air or light.
What is the molecular weight of silver bisulfate?
Name: Silver Hydrogen Sulfate. Alias: Silver Bisulfate. Formula: AgHSO4. Molar Mass: 204.9387.
How is hydrogen sulfide and silver sulfide related?
The silver ion and the sulfide ion stick together to form silver sulfide, Ag 2 S. Silver sulfide is generated when elemental silver reacts with hydrogen sulfide gas, which is present in air. Hydrogen sulfide gas is generated by decomposing plants and animals. Silver sulfide is generally a compound that doesn’t have many beneficial uses.