What is the full formula of silver nitrate?
AgNO3
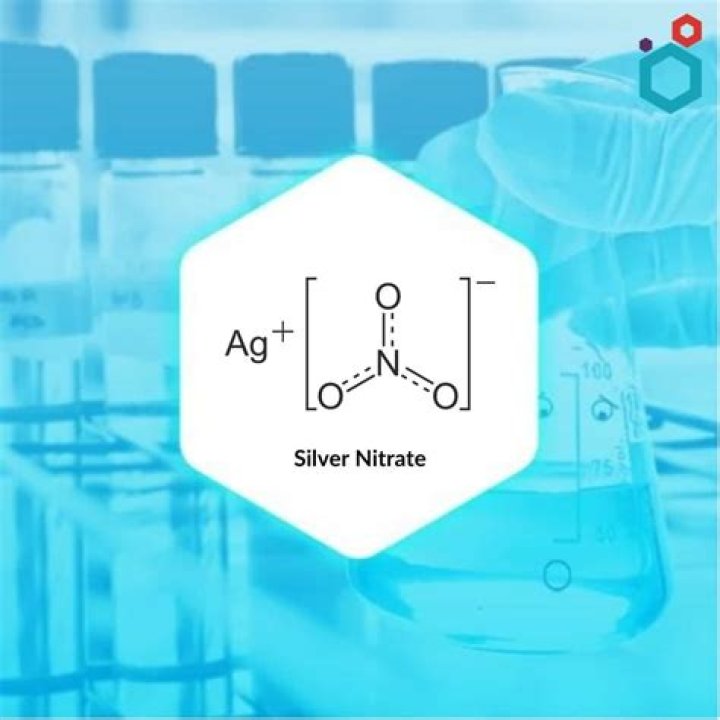
Silver nitrate is an inorganic compound with chemical formula AgNO3.
What element is AgNO3?
Silver nitrate is a chemical compound with the formula AgNO3. It consists of an ionic bond between the silver cation (Ag+) and the nitrate anion (NO3–). Due to the ionic nature of this compound, it readily dissolves in water and dissociates into its constituent ions.
How do you make AgNO3?
Silver nitrate can be prepared by reacting silver, such as silver bullion or silver foil, with nitric acid, resulting in silver nitrate, water, and oxides of nitrogen. Reaction byproducts depend upon the concentration of nitric acid used.
What is AgNO3 used for?
Silver nitrate is a natural compound that is used as an antiinfective agent. Silver nitrate topical (for use on the skin) is used to cauterize infected tissues around a skin wound. Silver nitrate can also help create a scab to help stop bleeding from a minor skin wound.
What is the oxidation state of N in AgNO3?
Hence, the oxidation number of Ag is +1 and NO3−has, N with +5 and O with -2 oxidation states in AgNO3. Note: The oxidation state of any atom is calculated in its ion form and not for the molecule, hence individual oxidation numbers are calculated. Here nitrogen is in its highest group oxidation number which is +5.
What can dissolve silver?
The full dissolution of silver requires nitric acid, or HNO3, which reacts with silver to form silver nitrate, a water-soluble compound. Put on rubber gloves pour 2 ounces of distilled water into a glass measuring cup. Add 1 ounce of concentrated nitric acid to the distilled water to bring the total volume to 3 oz.
What is the oxidation number of silver ion in silver nitrate?
+1
Silver nitrate is a chemical compound. Its chemical formula is AgNO3. It contains silver in its +1 oxidation state as an ion. It also has nitrate.
Which is the correct formula for silver nitrate?
Silver nitrate is an inorganic compound with the chemical formula AgNO3. In its solid form, silver nitrate is coordinated in a trigonal planar arrangement. It is often used as a precursor to other silver -containing compounds.
How much does one mole of silver nitrate weigh?
Total: 169.8731 grams. 1 Mole of Silver Nitrate. 1 Mole of Silver. 1 Mole of Nitrogen. 3 Moles of Oxygen [ Note: 1 Mole of Oxygen = 15.9994 grams (about 13.5 Cubic Feet!) ] From the table above, we can see that 1 Mole of Silver Nitrate weighs about 169.87 grams.
What can silver nitrate be used to prepare?
Silver nitrate is a precursor of many other silver compounds that are used. Hence, one of the use of Silver Nitrate is in the preparation of other Silver compounds. Silver nitrate is a very caustic chemical. This is used in the preparation of other silver salts.
What is the reaction between silver nitrate and ethanol?
The hazards of AgNO 3 include its toxic and corrosive nature. The reaction between silver nitrate and ethanol is explosive. The silver present in this compound is displaced by copper, which forms copper nitrate. The chemical equation for this reaction is given by 2AgNO 3 + Cu → Cu(NO 3) 2 + 2Ag.