What is the mass of one silver?
Thus, Mass of atoms of silver is 108 g.
What is the molar mass of silver in g mol?
107.9 g/mol
The molar mass of silver is 107.9 g/mol.
What is the mass of silver 107?
106.9 amu
Silver has two naturally occurring isotopes. Ag-107 has an abundance of 51.82% and mass of 106.9 amu. Ag-109 has a relative abundance of 48.18% and a mass of 108.9 amu.
What is the formula of atomic mass?
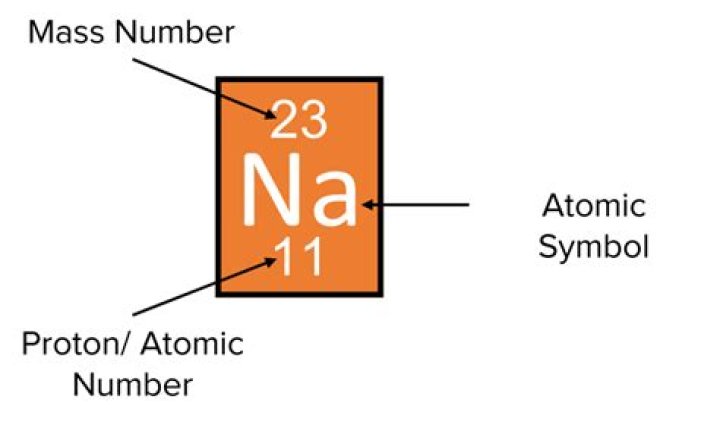
Together, the number of protons and the number of neutrons determine an element’s mass number: mass number = protons + neutrons. If you want to calculate how many neutrons an atom has, you can simply subtract the number of protons, or atomic number, from the mass number.
How do you find the mass?
Mass is always constant for a body. One way to calculate mass: Mass = volume × density. Weight is the measure of the gravitational force acting on a mass. The SI unit of mass is “kilogram”.
How many moles are in 21 grams of lithium?
How many grams Lithium in 1 mol? The answer is 6.941.
How is silver-107 the same as silver 109?
Naturally occurring silver (47Ag) is composed of the two stable isotopes 107Ag and 109Ag in almost equal proportions, with 107Ag being slightly more abundant (51.839% natural abundance). Isotopes of silver range in atomic weight from 92.950 u (93Ag) to 129.950 u (130Ag).
How do you find the abundance of silver-107?
The atomic mass of an element is the weighted average of the atomic masses of its isotopes. That means that we multiply the atomic mass of each isotope by a number representing its relative importance (that is, its percent abundance). is 100 % – 51.82 % = 48.18 %. Let x represent the atomic mass of Ag-107.
Where is the atomic mass?
Mass Number Experimental data showed that the vast majority of the mass of an atom is concentrated in its nucleus, which is composed of protons and neutrons. The mass number (represented by the letter A) is defined as the total number of protons and neutrons in an atom.
What is the mass of 1 atom of silver?
The molar mass of silver (Ag) is 107.87 g/mol (atomic weight in g/mol). Use dimensional analysis to calculate the mass of 1 atom of Ag from its molar mass and Avogadro’s number of atoms. 107.87 g/mol × (1 mol Ag/6.022×10^23 atoms Ag) = 1.791×10^-22 g/atom to four significant figures 1 atom Ag has a mass of ~ 1.791×10^-22 g. .
How to find the mass of an iron atom?
To find the mass of 1 atom of iron, you know that 1 atom of iron = 55.845 atomic mass units/amu. One amu is equal to 1.66 x 10-27 kg, (commonly, 1.66 x 10-27 g). Set up your equation so that the units cancel out.
How do you calculate the mass of 1 GM?
you must be thinking that 1 gm is weight but it is really mass.weight is a force that is measured by a weighing machine but it gives you the mass after dividing it by 9.8 or 10. Woah ! You just answered your question yourself. The mass is 1 gm itself. Besides, if you want to calculate its weight, then its given by the below formula, Say, g = 9.8
What are the units of mass in SI?
The units of mass are kg, gm and lb (pound) in the SI, CGS and imperial systems respectively. Weight is the force with which a body is attracted to the center of the earth.