What materials were used in the gold-foil experiment?
For the metal foil, they tested a variety of metals, but they preferred gold because they could make the foil very thin, as gold is very malleable. As a source of alpha particles, Rutherford’s substance of choice was radon, a substance several million times more radioactive than uranium.
What two main materials did Rutherford use in his gold-foil experiment?
In 1899 Ernest Rutherford studied the absorption of radioactivity by thin sheets of metal foil and found two components: alpha (a) radiation, which is absorbed by a few thousandths of a centimeter of metal foil, and beta (b) radiation, which can pass through 100 times as much foil before it was absorbed.
What did Rutherford bombard the gold foil with?
alpha particles
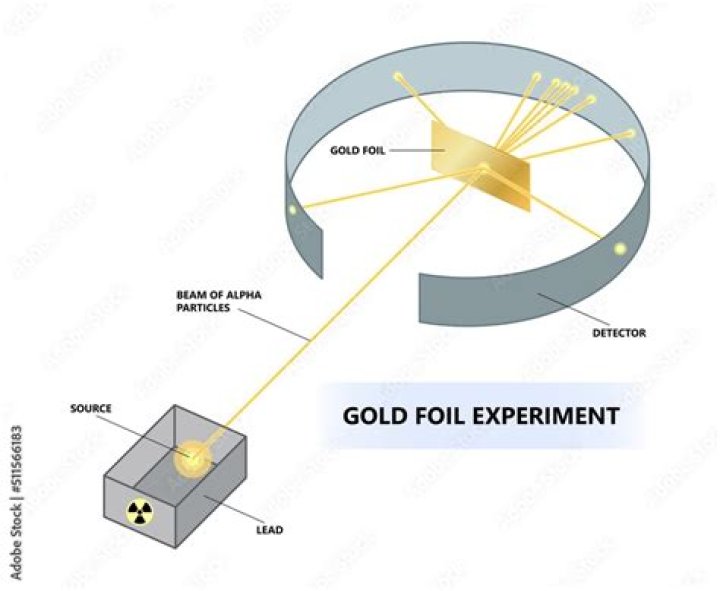
The Gold Foil Experiment. In 1911, Rutherford and coworkers Hans Geiger and Ernest Marsden initiated a series of groundbreaking experiments that would completely change the accepted model of the atom. They bombarded very thin sheets of gold foil with fast moving alpha particles.
What is the conclusion of Rutherford’s gold foil experiment?
Observation And Conclusion Of Rutherford’s Scattering Experiment
| Observations | Conclusion |
|---|---|
| Few particles bounce off the nucleus | Indicates that all the positive charge and mass of the gold atom were concentrated in a very small volume within the atom. |
What kind of foil was used in Rutherford’s experiment?
In this experiment, Rutherford used Gold Foil which was extremely thin sheet, not more than 1000 atoms thick. Gold Foil was bombarded with positively charged α-particles.
What was the purpose of the gold foil experiment?
Rutherford’s gold foil experiment (Rutherford’s alpha particle scattering experiment) refers to an experiment carried out by Ernest Rutherford, Hans Geiger, and Ernest Marsden at the University of Manchester in the early 1900s. In the experiment, Rutherford and his two students studied how alpha particles fired at a thin piece …
What kind of particles are in gold foil?
They bombarded a thin gold foil of thickness approximately 8.6 x 10 -6 cm with a beam of alpha particles in a vacuum. Alpha particles are positively charged particles with a mass of about four times that of a hydrogen atom and are found in radioactive natural substances.
Why was gold used in the Rutherford experiment?
Ans. Rutherford needed the thinnest sheet of metal for performing the experiment. Gold is most malleable compared to all other metals and was therefore used for the experiment.