What metals are more reactive than tin?
| Substance | Comment |
|---|---|
| Zinc | Metal: fairly reactive |
| Iron | Metal: not very reactive |
| Tin | Metal: not very reactive |
| Lead | Metal: not very reactive |
Is silver more reactive than metal?
In a reactivity series, the most reactive element is placed at the top and the least reactive element at the bottom. More reactive metals have a greater tendency to lose electrons and form positive ions ….The reactivity series.
| Element | Reaction with dilute acids |
|---|---|
| Silver | Barely reacts |
| Gold | Does not react |
Is tin more reactive than aluminum?
Tin and lead are both less active than aluminum. Neither metal reacts with either dilute hydrochloric acid or dilute sulfuric acid at room temperature. Tin, when heated, reacts with either concentrated hydrochloric acid or concentrated sulfuric acid.
Which metal is less reactive than tin?
As we can see that in this option, the first metal that is copper is below that hydrogen in the reactivity series so, copper is less reactive than hydrogen and the second metal that is tin is placed above the hydrogen in the reactivity series, so tin is more reactive than hydrogen.
Is tin a reactive metal?
Tin is more reactive than germanium, which is immediately above tin in the periodic table. It is stable in air under ambient conditions but on heating in air or oygen, tin reacts with oxygen to from tin dioxide, SnO2.
Which is the least reactive metal?
Platinum
Calcium. Hint: Platinum is one of the rare metals on earth and is also called the noble metal as it is not found in combined states in natural surroundings. Complete answer: Platinum is the least reactive of all the options.
How will you show that silver is less reactive than copper?
Silver is chemically less reactive than copper can be shown by displacement reaction. If a piece of silver is placed in a solution of CuSO4 no reaction will take place as Ag is less reactive than Cu and can’t replace Cu from CuSO4 solution. This shows Ag is chemically less reactive than copper.
What is the least reactive metal?
What metal is the least reactive?
gold
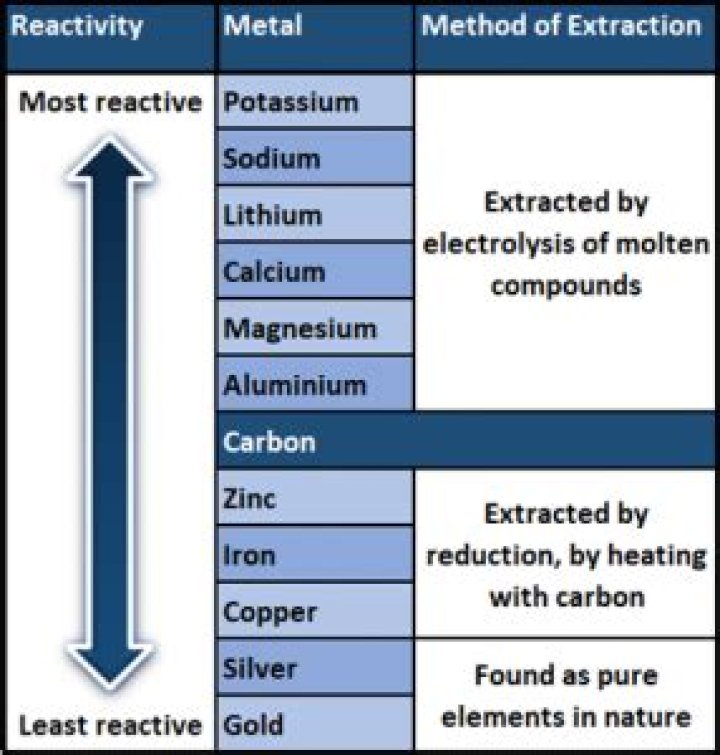
The reactivity series lists the metals in order of reactivity, from the most reactive at the top (potassium) to the least reactive at the bottom (gold).
What is tin reactive to?
Tin is more reactive than germanium, which is immediately above tin in the periodic table. It is stable to water under ambient conditions but on heating with steam, tin reacts with water to from tin dioxide, SnO2 and hydrogen.
Which is not reactive silver or gold metal?
Silver Metal: not reactive Gold Metal: not reactive Platinum Metal: not reactive
Which is the most reactive metal in the world?
Some metals are more reactive than others. We put then in order of their reactivity in a series that starts with the most reactive. This is the Reactivity Series. Substance Symbol Comment Potassium Metal: very reactive
How can we see the reactivity of metals?
We can examine the reactivity of metals by observing their reactions with oxygen, water, steam and whether it displaces other metals in displacement reactions. This table summarises the reactions of some metals in the reactivity series. Hydrogen is included for comparison.
What makes copper and silver react with nitric acid?
Copper and silver will react with nitric acid; but because nitric acid is an oxidizing acid, the oxidizing agent is not the H + ion as in normal acids, but the NO 3− ion. The reactivity series is sometimes quoted in the strict reverse order of standard electrode potentials, when it is also known as the ” electrochemical series ” :-