What scientist used the gold foil test?
Ernest Rutherford
Ernest Rutherford and the gold foil experiment In his famous gold foil experiment, Rutherford fired a thin beam of α particles (pronounced alpha particles) at a very thin sheet of pure gold.
Who designed and carried out the gold foil experiment?
In 1905, Ernest Rutherford did an experiment to test the plum pudding model. His two students, Hans Geiger and Ernest Marsden, directed a beam of alpha particles at a very thin gold leaf suspended in a vacuum . Alpha particles are a form of nuclear radiation with a large positive charge.
What is the gold foil experiment name the scientist who performed this experiment write the conclusions and shortcomings of Rutherford’s model of atom 3m?
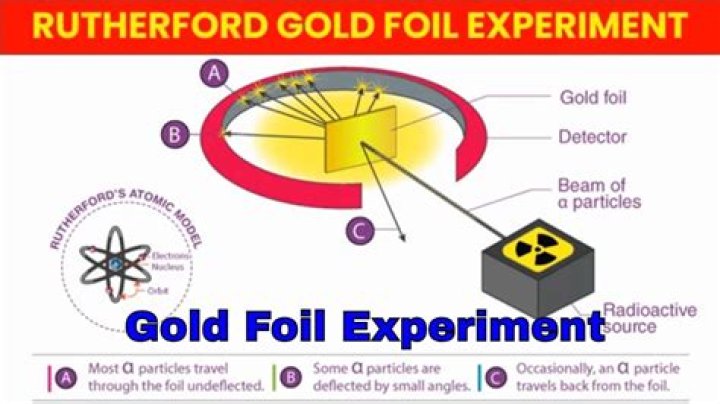
In 1911, Rutherford performed the gold foil experiment. He bombarded a stream of a-particles on a gold foil, a thin sheet which was 0.00006 cm thick in an evacuated chamber. An a-particle is a positively charged helium ion (He +). Most of the a-particles passed straight through the foil without any deflection.
Why is it called the gold foil experiment?
Rutherford’s experiment is called the gold foil experiment because he used gold foil. 3. How did he know that an atom was mostly empty space? He knew that an atom was made of mostly empty space because most particles passed straight through the foil.
Who was involved in the gold foil experiment?
Gold Foil Experiment Who did the Gold Foil Experiment? The gold foil experiment was a pathbreaking work conducted by scientists Hans Geiger and Ernest Marsden under the supervision of Nobel laureate physicist Ernest Rutherford that led to the discovery of the proper structure of an atom.
What did Rutherford do with the gold foil?
Rutherford’s Gold Foil Experiment Animation Summary Rutherford demonstrated his experiment on bombarding thin gold foil with alpha particles contributed immensely to the atomic theory by proposing his nuclear atomic model.
How did Rutherfords gold foil experiment disprove the plum pudding model?
How did Rutherford’s gold foil experiment disprove the plum pudding model? Chemistry A Closer Look at The Atom Rutherford’s Gold Foil Experiment.
What did Geiger and Marsden observe in the foil experiment?
Left: Had Thomson’s model been correct, all the alpha particles should have passed through the foil with minimal scattering. Right: What Geiger and Marsden observed was that a small fraction of the alpha particles experienced strong deflection.