What type of bond is copper and copper?
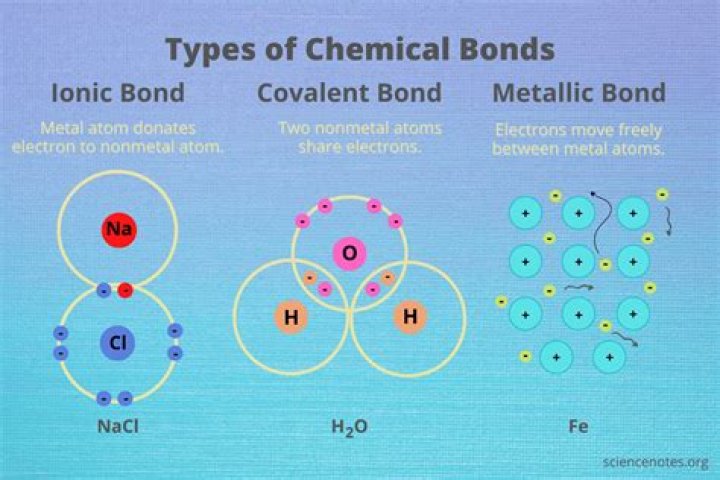
Explanation: Copper wire consists of Cu nuclei (that are positively charged due to the positively charged protons) that are surrounded by a “sea” of delocalized electrons (that are negatively charged that move freely throughout the material which is known as metallic bonding.
What type of covalent bond is formed in nitrogen molecule?
Answer: Nitrogen atoms will form three covalent bonds (also called triple covalent) between two atoms of nitrogen because each nitrogen atom needs three electrons to fill its outermost shell. Another example of a nonpolar covalent bond is found in the methane (CH4) molecule.
What type of bond is formed between copper and zinc?
In the case of an alloy of copper and zinc, you have what we call brass, and the bond that is present is a metallic bond. How do we know? Because that’s the definition of a metallic bond. It is a highly delocalized sea of electrons and positively charged metal ions.
Is copper ionic or covalent?
Most metal elements/atoms lose electrons when they form the ionic bonds with other elements. However, copper is unique as it can form two ionic bonds.
What can bond with copper?
Cyanoacrylate adhesives or instant adhesives are ideal for bonding copper. The highest strength bonds are created with methyl cyanoacrylates such as the original Permabond 910.
What type of bond is solid copper?
Metallic bonds occur among metal atoms. Whereas ionic bonds join metals to non-metals, metallic bonding joins a bulk of metal atoms. A sheet of aluminum foil and a copper wire are both places where you can see metallic bonding in action.
Can nitrogen form 4 bonds?
If you look at the above image you can see that when nitrogen has a positive charge (one less electron), it can form four covalent bonds. Either with single, double, or triple bonds. It is similar to phosphorus in this regard because they both have five valence electrons (four when they have a positive charge).
What type of bonds are in glucose?
The carbon atoms in the glucose ring each have four covalent bonds. The best, or optimum angle, between all these bonds is 109.5o, which results in a perfect tetrahedron.
What kind of bond is CR?
The covalent bonds form between two or more NON-METAL elements. These bonds are typically strong and flexible….VIDEO Explanation of bonding examples 1.
| Na to Br | Ionic |
|---|---|
| Ca to S | Ionic |
| Si to Cl | Covalent |
| F to F | Covalent |
| Sr to Cr | Metallic |
Does copper have covalent bond?
All bonds, and particularly Cu–S, are highly covalent in character.
What kind of bond is copper II hydroxide?
Copper(ii)–hydroxide facilitated C–C bond formation: the carboxamido pyridine system versus the methylimino pyridine system†
What are 3 interesting facts about copper?
10 Copper Facts
- Copper has a reddish-metallic coloring that is unique among all the elements.
- Copper was the first metal to be worked by man, along with gold and meteoritic iron.
- Copper is an essential element for human nutrition.
- Copper readily forms alloys with other metals.
- Copper is a natural antibacterial agent.
Is copper found uncombined?
Copper was one of the earliest elements known to man. At one time, it could be found lying on the ground in its native state or uncombined state. Copper is a transition metal, one of several elements found in rows 4 through 7 between Groups 2 and 13 in the periodic table.
Is a hydrogen bond?
A hydrogen bond is the electromagnetic attraction created between a partially positively charged hydrogen atom attached to a highly electronegative atom and another nearby electronegative atom. A hydrogen bond is a type of dipole-dipole interaction; it is not a true chemical bond.
Why are metallic bonds not soluble in water?
Metallic bonds are not soluble in water because: They are held together by strong metallic bonds and so no solvent to solute attractions could be stronger than these, so these substances are insoluble also they don’t have the necessary intermolecular forces (namely hydrogen bonds) that are present in water.
Which types of bonds are the strongest?
Covalent bonds are the strongest (*see note below) and most common form of chemical bond in living organisms. The hydrogen and oxygen atoms that combine to form water molecules are bound together by strong covalent bonds.
Why nitrogen can form only 4 bonds?
By sharing the three 2p electrons, nitrogen can form three covalent bonds. But still the nitrogen atom has a lone pair of electrons form 2s orbital. By donating these two electrons from lone pair, it can form one bond. For example, NH4+.
Why nitrogen can form 5 bonds?
Nitrogen needs 5 unpaired electrons to form 5 covalent bonds, but there are only 4 orbitals ( one s and three p) in its valence shell so there has to be a pair of electrons in one of the orbitals giving only 3 unpaired electrons.