What was surprising about the results of the gold foil experiment?
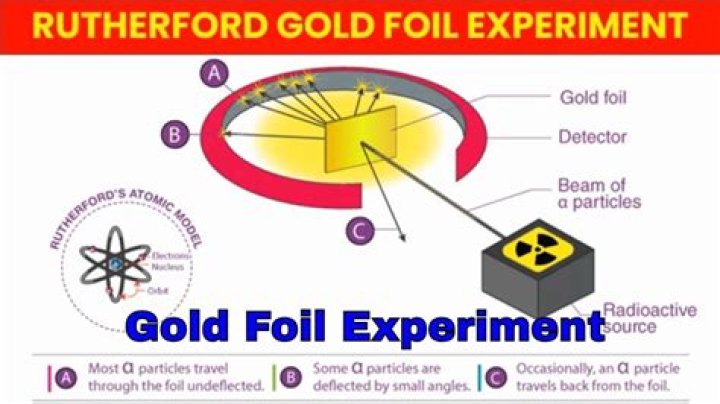
Rutherford’s gold foil experiment showed that the atom is mostly empty space with a tiny, dense, positively-charged nucleus. Based on these results, Rutherford proposed the nuclear model of the atom.
What was surprising in Rutherford’s experiment?
Rutherford discovered the proton, and he also discovered that the atom is mainly empty space. He noticed that a beam of alpha particles was scattered back where it came from by gold atoms, and, since it was known that alpha particles were positive, it was deduced that there was a dense positive core in the nucleus.
Which was not a conclusion of the gold foil experiment?
It was not a conclusion because it is neutral. It is not positive nor negative.
What did Rutherford do with the gold foil?
Rutherford’s Gold Foil Experiment Animation Summary Rutherford demonstrated his experiment on bombarding thin gold foil with alpha particles contributed immensely to the atomic theory by proposing his nuclear atomic model.
What was the purpose of the gold foil experiment?
Rutherford’s gold foil experiment (Rutherford’s alpha particle scattering experiment) refers to an experiment carried out by Ernest Rutherford, Hans Geiger, and Ernest Marsden at the University of Manchester in the early 1900s. In the experiment, Rutherford and his two students studied how alpha particles fired at a thin piece …
How did Rutherfords gold foil experiment disprove the plum pudding model?
How did Rutherford’s gold foil experiment disprove the plum pudding model? Chemistry A Closer Look at The Atom Rutherford’s Gold Foil Experiment.
What was the result of the Rutherford experiment?
The Rutherford Experiment. Because alpha particles have about 8000 times the mass of an electron and impacted the foil at very high velocities, it was clear that very strong forces were necessary to deflect and backscatter these particles. Rutherford explained this phenomenon with a revitalized model of the atom in which most…