What was the gold foil experiment What did it discover?
The gold-foil experiment showed that the atom consists of a small, massive, positively charged nucleus with the negatively charged electrons being at a great distance from the centre.
Who did the gold foil experiment and what did they discover from it?
In 1899 Ernest Rutherford studied the absorption of radioactivity by thin sheets of metal foil and found two components: alpha (a) radiation, which is absorbed by a few thousandths of a centimeter of metal foil, and beta (b) radiation, which can pass through 100 times as much foil before it was absorbed.
What scientist is responsible for the gold foil experiment?
Rutherford’s nuclear model. Rutherford overturned Thomson’s model in 1911 with his famous gold-foil experiment, in which he demonstrated that the atom has a tiny, massive nucleus.
Who discovered the nucleus with his famous gold foil experiment?
Ernest Rutherford
Ernest Rutherford and the gold foil experiment The next groundbreaking experiment in the history of the atom was performed by Ernest Rutherford, a physicist from New Zealand who spent most of his career in England and Canada.
What was Rutherford’s experiment called?
the gold foil experiment
Rutherford’s experiment is called the gold foil experiment because he used gold foil.
What was Rutherford’s experiment?
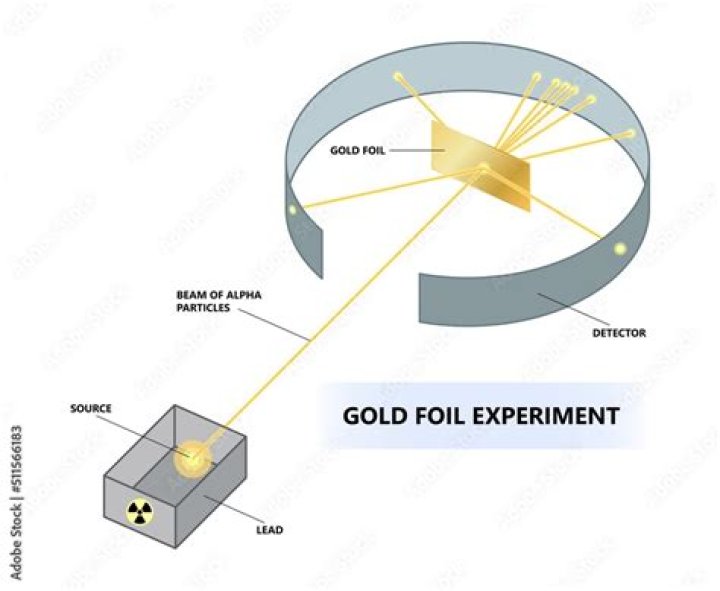
Ernest Rutherford’s most famous experiment is the gold foil experiment. A beam of alpha particles was aimed at a piece of gold foil. Most alpha particles passed through the foil, but a few were scattered backward. This showed that most of the atom is empty space surrounding a tiny nucleus.
Why did the gold foil have to be thin?
Gold was used because it was the only metal that could be rolled out to be very, very thin without cracking. Since the gold foil was very thin, it was thought that the alpha particles could pass straight through it, or possibly puncture the foil. most of the alpha particles did pass straight through the foil.
What was the conclusion of Rutherford’s experiment?
Observation And Conclusion Of Rutherford’s Scattering Experiment
| Observations | Conclusion |
|---|---|
| Few particles bounce off the nucleus | Indicates that all the positive charge and mass of the gold atom were concentrated in a very small volume within the atom. |
What did Rutherford’s gold foil experiment tell about the atom?
Most of the time, the alpha particles would pass through the foil without any change in their trajectories, which is what was expected if JJ Thomson’s plum pudding model of the atom was correct.
Who was the first scientist to discover the nucleus?
The Geiger–Marsden experiment(s) (also called the Rutherford gold foil experiment) were a landmark series of experiments by which scientists discovered that every atom contains a nucleus where all of its positive charge and most of its mass are concentrated.
What happens when gold foil is hit with alpha particles?
A piece of gold foil was hit with alpha particles, which have a positive charge. Most alpha particles went right through. This showed that the gold atoms were mostly empty space. Some particles had their paths bent at large angles. A few even bounced backward.
How did Rutherford prove Thomson’s model of the atom wrong?
In a single famous experiment, however, Rutherford showed unambiguously that Thomson’s model of the atom was incorrect. Rutherford aimed a stream of α particles at a very thin gold foil target (Figure 2.4. 7 a) and examined how the α particles were scattered by the foil.