What was the resulting conclusion of the gold foil experiment?
What conclusion was a direct result of the gold foil experiment? The gold foil experiment led to the conclusion that each atom in the foil was composed mostly of empty space because most alpha particles directed at the foil 1) An atom is mostly empty space with a dense, positively charged nucleus.
What did the gold foil experiment determine about the atom?
The gold-foil experiment showed that the atom consists of a small, massive, positively charged nucleus with the negatively charged electrons being at a great distance from the centre.
What is the conclusion of Rutherford experiment?
Observation And Conclusion Of Rutherford’s Scattering Experiment
| Observations | Conclusion |
|---|---|
| Few particles bounce off the nucleus | Indicates that all the positive charge and mass of the gold atom were concentrated in a very small volume within the atom. |
What three conclusions came from the gold foil experiment?
Most of the alpha particles did pass straight through the foil. The atom being mostly empty space. A small number of alpha particles were deflected by large angles (> 4°) as they passed through the foil. There is a concentration of positive charge in the atom.
Who discovered neutrons?
James Chadwick
In May 1932 James Chadwick announced that the core also contained a new uncharged particle, which he called the neutron. Chadwick was born in1891 in Manchester, England.
Which is true about the atomic number?
The atomic number uniquely identifies a chemical element. It is identical to the charge number of the nucleus. In an uncharged atom, the atomic number is also equal to the number of electrons. The sum of the atomic number Z and the number of neutrons N gives the mass number A of an atom.
What were two conclusions of the gold foil experiment?
From the location and number of α-particles reaching the screen, Rutherford concluded the following: i) Almost 99% of the α-particles pass through the gold foil without any deflection. So atom must be having a lot of empty space in it. ii) Several α-particles get deflected at angles.
Why was Rutherford’s experiment important?
Rutherford’s experiment showed that atoms consisted of a dense mass which was surrounded by mostly empty space – the nucleus! The conclusion that could be formed from this result was that atoms had an inner core which contained most of the mass of an atom and was positively charged.
What were the observations of Rutherford’s experiment?
His two primary observations were: Most α particles passed straight through the gold foil, which showed that atoms are mostly empty space. Some of the α particles were deflected at various angles, and sometimes even back at the radioactive source.
How does an atom pass through gold foil?
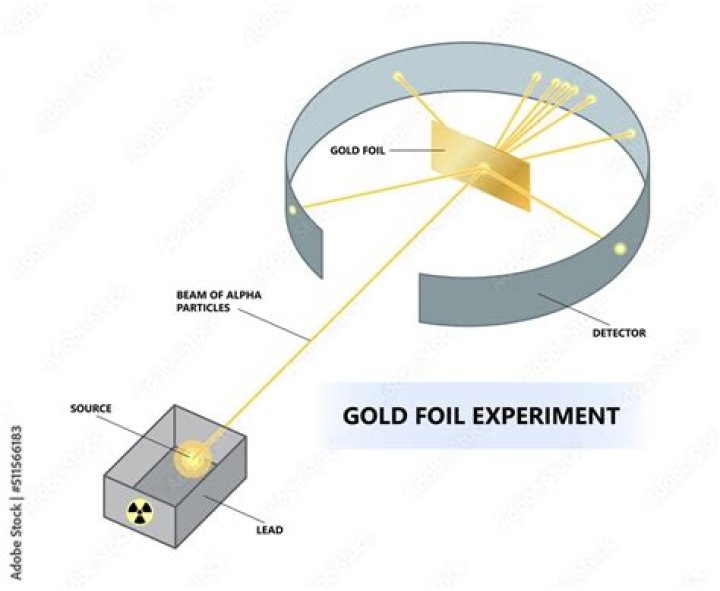
What is the description of the gold foil experiment?
What is the Gold Foil Experiment. Description. The scientists bombarded a thin gold foil of thickness approximately 8.6 x 10-6 cm with a beam of alpha particles in vacuum. They used gold since it is highly malleable, producing sheets that can be only a few atoms thick, thereby ensuring smooth passage of the alpha particles.
What was the conclusion of the Rutherford experiment?
Experiment Conclusion: 1 Most of the space inside an atom is empty space. 2 The most mass of the atom (which are protons and neutrons) are concentrated at the center of the atom which can deflect alpha particles at larger angles. 3 The atom can be depicted as a tennis ball with 5 km radius.
What did Rutherford do with the gold foil?
Rutherford’s Gold Foil Experiment Animation Summary Rutherford demonstrated his experiment on bombarding thin gold foil with alpha particles contributed immensely to the atomic theory by proposing his nuclear atomic model.