Where does the rusting of iron take place?
It means that rusting of iron can take place in ordinary water also. Rusting of iron takes place in both ordinary and distilled water, which is option ‘c’. Additional Information: The other factor on which rusting of iron depends is humidity. As iron requires oxygen in air, water or water vapours for rust.
What happens when rusting of iron takes place?
The oxygen atoms bond with iron atoms, resulting in the formation of iron oxides. This weakens the bonds between the iron atoms in the object/structure. The reaction of the rusting of iron involves an increase in the oxidation state of iron, accompanied by a loss of electrons.
What is iron rust and how is it formed?
Rust is an iron oxide, a usually reddish-brown oxide formed by the reaction of iron and oxygen in the catalytic presence of water or air moisture. Rust consists of hydrous iron(III) oxides (Fe2O3·nH2O) and iron(III) oxide-hydroxide (FeO(OH), Fe(OH)3), and is typically associated with the corrosion of refined iron.
What are the three types of rust?
Types of rust:
- Yellow Rust. Iron oxide-hydroxide FeO(OH)H2O (high moisture) Yellow rust is present in metals that are found in large amounts of standing water.
- Brown Rust. Oxide Fe2O3 (high oxygen/low moisture)
- Black Rust. Iron (II)oxide – Fe3O4 (limited oxygen)
How can rusting of iron be prevented?
9 Ways to Prevent Rust
- Use an Alloy. Many outdoor structures, like this bridge, are made from COR-TEN steel to reduce the effects of rust.
- Apply Oil.
- Apply a Dry Coating.
- Paint the Metal.
- Store Properly.
- Galvanize.
- Blueing.
- Powder Coating.
Why does the iron turn yellowish brown?
Organic Iron and Tannins Iron can combine with different naturally-occurring organic acids or tannins. Organic iron occurs when iron combines with an organic acid. Water with this type of iron is usually yellow or brown, but may be colorless.
Does rust continue when dry?
Will rust spread if kept dry? Rust does not spread through contact like a biological infection. This means that if one part of the piece is exposed to water, oxygen, and electrolytes but the rust of the piece is kept clean and dry, the protected metal will not rest at the rate of the wet metal.
What are the stages of rust?
Rust fungi may produce as many as five different stages in their life cycles: Stage 0: Spermogonia bearing spermatia (sexual recombination occurs here) Stage I: Aecia bearing aeciospores Stage II: Uredinia bearing urediniospores (summer, or repeating spores) Stage III: Telia bearing teliospores Stage IV: …
What causes rust on the surface of iron?
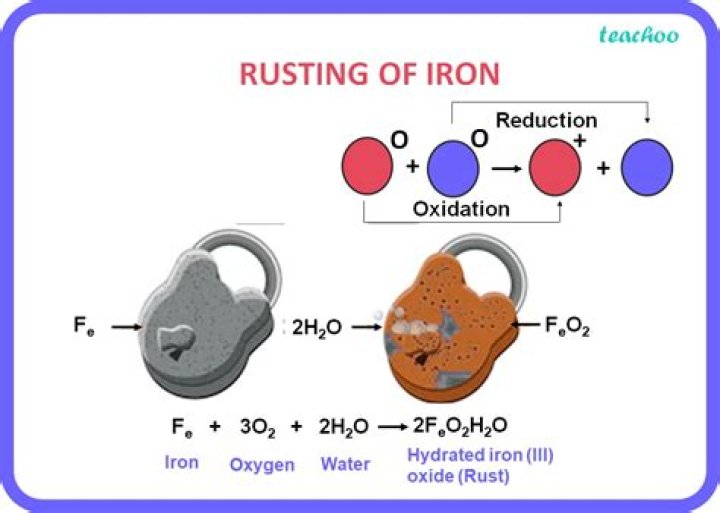
Rusting of Iron. When a piece of iron is left out in the open for a while, a film of brownish substance gets deposited on its surface called rust. Rusting occurs when iron is exposed to oxygen in air and water or water vapor. Rust is Iron oxide. Iron plus oxygen plus water vapor gives iron oxide.
Where does rust occur in the oxidation reaction?
Although rust is considered the result of an oxidation reaction, it’s worth noting not all iron oxides are rust. Rust forms when oxygen reacts with iron but simply putting iron and oxygen together isn’t sufficient. Although about 20 percent of air consists of oxygen, rusting doesn’t occur in dry air. It occurs in moist air and in water.
What is the name of the chemical that causes rust?
Key Takeaways: How Rust Works. Rust is the common name of the chemical called iron oxide. Technically, it’s iron oxide hydrate, because pure iron oxide isn’t rust. Rust forms when iron or its alloys are exposed to moist air. The oxygen and water in air react with the metal to form the hydrated oxide.
What do you need to know about rust and steel?
Rust is brittle, fragile, progressive, and weakens iron and steel. To protect iron and its alloys from rust, the surface needs to be separated from air and water. Coatings can be applied to iron. Stainless steel contains chromium, which forms an oxide, much like how iron forms rust.