Which compound is ionic?
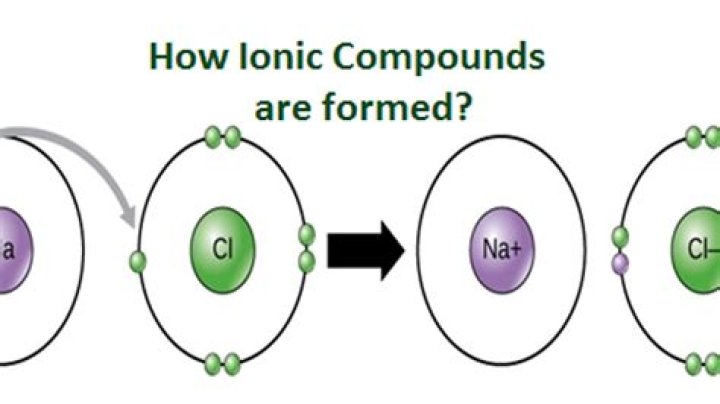
Ionic Compounds Are Balanced Table salt is an example of an ionic compound. Sodium and chlorine ions come together to form sodium chloride, or NaCl. The sodium atom in this compound loses an electron to become Na+, while the chlorine atom gains an electron to become Cl-.
What type of compound is magnesium chloride?
inorganic compound
Magnesium chloride. An inorganic compound consisting of one magnesium and two chloride ions. The compound is used in medicine as a source of magnesium ions, which are essential for many cellular activities. It has also been used as a cathartic and in alloys.
Is mgcl2 a compound?
Magnesium dichloride
Magnesium chloride/IUPAC ID
How do you identify an ionic compound?
To find the formula of an ionic compound, first identify the cation and write down its symbol and charge. Then, identify the anion and write down its symbol and charge. Finally, combine the two ions to form an electrically neutral compound.
What is ionic compound explain with example?
Ionic compounds are those compounds which are formed by the transfer of electrons from a metal to a non-metal. For example, NaCl. (i) Physical nature: Ionic compounds are hard and solid due to strong force of attraction between oppositely charged ions.
What are the 10 examples of compound?
Examples of Compounds – Common Compounds
- C2H6O: Alcohol. NaCl: Salt. C2H6O: Ethanol. C2H4O2: Vinegar. NH3: Ammonia. C2H4O2: Acetic Acid. C4H10: Butane.
- H2SO4 : Sulfuric Acid. CH4: Methane.
- C12H22O11: Sucrose. C3H8: Propane.
- NaHCO3: Baking Soda. N2O: Nitrogen. C6H8O7: Citric Acid. C8H18: Octane. C10H16O: Camphor.
What are 2 examples of mixtures?
Few Examples of Mixtures We Find in Our Daily Lives.
- Sand and water.
- Salt and water.
- Sugar and salt.
- Ethanol in water.
- Air.
- Soda.
- Salt and pepper.
- Solutions, colloids, suspensions.
What are the benefits of magnesium chloride?
This medication is a mineral supplement used to prevent and treat low amounts of magnesium in the blood. Some brands are also used to treat symptoms of too much stomach acid such as stomach upset, heartburn, and acid indigestion.
Is magnesium chloride harmful?
Magnesium chloride supplements are considered safe if used as directed. Common side effects include stomach upset, nausea, diarrhea, and vomiting. Many of these side effects can be alleviated by taking the supplement with food. Nearly all forms of magnesium supplements have a laxative effect.
How to tell if MgCl2 is ionic or covalent?
To tell if MgCl2 (Magnesium chloride) is ionic or covalent (also called molecular) we look at the Periodic Table that and see that Mg is a metal and Cl is a non-metal. When we have a metal and a non-metal the compound is usually considered ionic.
What kind of compound is magnesium chloride or MgCl2?
What kind of compound is Magnesium chloride or MgCl2 . . , Magnesium has 12 electrons, and its electronic configuration can be written as 2,8,2. Thus Magnesium has two electrons in its outermost orbit or it can be said that Magnesium has 2 valence electrons.
What kind of bond does magnesium and chlorine form?
1) Magnesium and chlorine form an ionic bond. Covalent bonds are formed when two or more atoms share electrons between them. Ionic bonds are when atoms gain or lose electrons to become charged species (ions) that share an electrostatic interaction called an ionic bond.
Which is a polar solvent, water or MgCl2?
We know that MgCl2 is an ionic solid, so we know that it is a polar compound since it is made up of ions. Water (H2O) is a polar solvent. The electronegativity difference between hydrogen and oxygen is about 1.4.