Which is least reactive metal copper or gold?
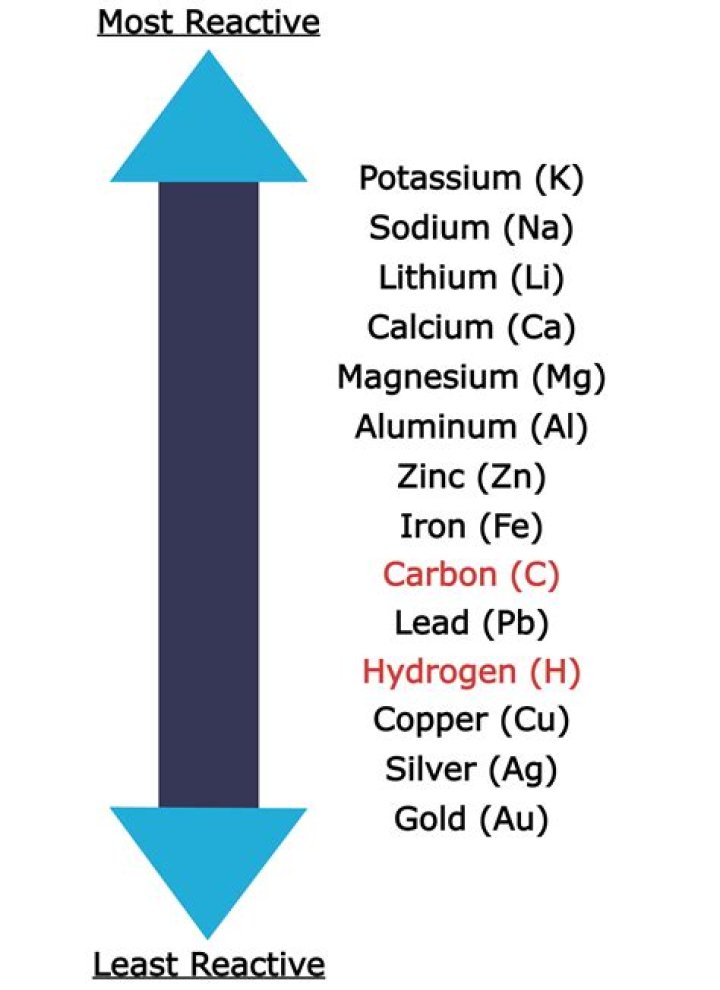
Complete step by step answer: We know that reactivity series is arranged in such a way that as we move from bottom to top, reactivity of the metals increases. As we can see in the series, out of all the given options gold is the least reactive metal. So, option D is correct.
Which is less reactive metal gold or silver?
The order of intensity of reactivity is known as reactivity series. Reactivity of element decreases on moving from top to bottom in the given reactivity series. In the reactivity series, copper, gold, and silver are at the bottom and hence are least reactive.
Is platinum less reactive than gold?
The more reactive the metal, the more rapid the reaction is. Platinum is placed below gold in the reactivity series.
Is gold the most non reactive metal?
THE unique role that gold plays in society is to a large extent related to the fact that it is the most noble of all metals: it is the least reactive metal towards atoms or molecules at the interface with a gas or a liquid.
Which metal is the least reactive metal?
Silver, gold, and platinum are metals with the least reactivity.
Why is gold the least reactive metal?
Since the 6s orbital with one electron is contracted, this electron is more tightly bound to the nucleus and less available for bonding with other atoms. The 4f and 5d orbitals expand, but can’t be involved in bond formation since they are completely filled. This is why gold is relatively unreactive.
Who is the least reactive metal?
Platinum
Platinum is the least reactive of all the options.
Which is more reactive metal or less reactive metal?
More reactive metals displace less reactive metals from their compounds and react with water. The reactivity series of metals is a chart listing metals in order of decreasing reactivity.
Why is gold less reactive than silver or copper?
Because they have various electrochemical potentials, various number of electronic shields and electrons and can be in compounds at various oxidation states. Gold electronic configuration for its outer electrons is 5d10 6s1. That means its 5d electrons are full, so it doesn’t have easily accessible d electrons or orbitals for reacting.
How does the reactivity series of metals work?
The reactivity series ranks metals by how readily they react. More reactive metals displace less reactive metals from their compounds and react with water. The reactivity series of metals is a chart listing metals in order of decreasing reactivity.
Why does gold not react in the reactivity series?
Gold. Does not react. Note that aluminium can be difficult to place in the correct position in the reactivity series during these experiments. This is because its protective aluminium oxide layer…