Which model of the atom was discovered through the gold foil experiment?
Rutherford’s nuclear model
atom: Rutherford’s nuclear model Rutherford overturned Thomson’s model in 1911 with his famous gold-foil experiment, in which he demonstrated……
Which of the following was supported by the gold foil experiment?
The model of the atom which was supported by the gold foil experiment was nuclear model.
What atomic model did Rutherford make?
Rutherford’s atomic model became known as the nuclear model. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom.
Why Rutherford model is called nuclear model?
Rutherford’s model of the atom is called the nuclear atom because it was the first atomic model to feature a nucleus at its core.
What was Rutherford’s theory?
Ernest Rutherford found that the atom is mostly empty space, with nearly all of its mass concentrated in a tiny central nucleus. The nucleus is positively charged and surrounded at a great distance by the negatively charged electrons.
What does the gold foil experiment tell us?
Rutherford’s gold foil experiment showed that the atom is mostly empty space with a tiny, dense, positively-charged nucleus. Based on these results, Rutherford proposed the nuclear model of the atom.
What is Bohr’s model called?
According to the Bohr model, often referred to as a planetary model, the electrons encircle the nucleus of the atom in specific allowable paths called orbits.
What are the two main features of Rutherford’s atomic model?
The salient features of this model are as follows: (i) The atom contains a central part called nucleus which is surrounded by electrons. (ii) The nucleus of an atom is positively charged. (iii) The size of the nucleus is very small as compared to the atomic size.
What was the purpose of the gold foil experiment?
How did Rutherford’s gold foil experiment explain the nuclear model?
The electrons revolve in circular orbits about a massive positive charge at the centre. His model explained why most of the α particles passed straight through the foil. The small positive nucleus would deflect the few particles that came close. The nuclear model replaced the plum pudding model.
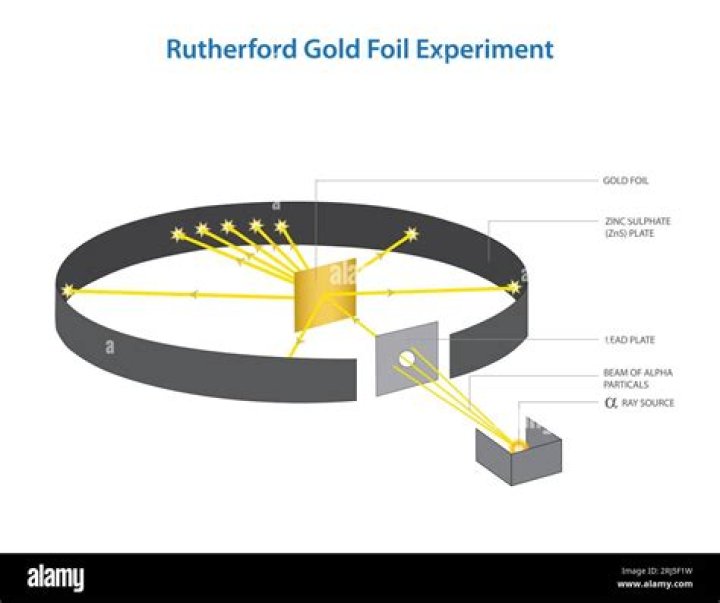
What is the description of the gold foil experiment?
What is the Gold Foil Experiment. Description. The scientists bombarded a thin gold foil of thickness approximately 8.6 x 10-6 cm with a beam of alpha particles in vacuum. They used gold since it is highly malleable, producing sheets that can be only a few atoms thick, thereby ensuring smooth passage of the alpha particles.
Why did Rutherford change the atom to a positive nucleus?
His model explained why most of the α particles passed straight through the foil. The small positive nucleus would deflect the few particles that came close. The nuclear model replaced the plum pudding model. The atom now consisted of a positive nucleus with negative electrons in circular orbits around it .
How are hydrogen atoms heavier than gold foil?
They are approximately 4 times heavier than Hydrogen atoms. Lead container acted as source for α-particles. Since he thought that the atom had a pudding-like structure, with electrons and positively charged particles embedded in it, he expected that, the α-particles would just pass through. Fig. 3 – Gold Foil Experiment