Who discovered protons using gold foil?
Rutherford
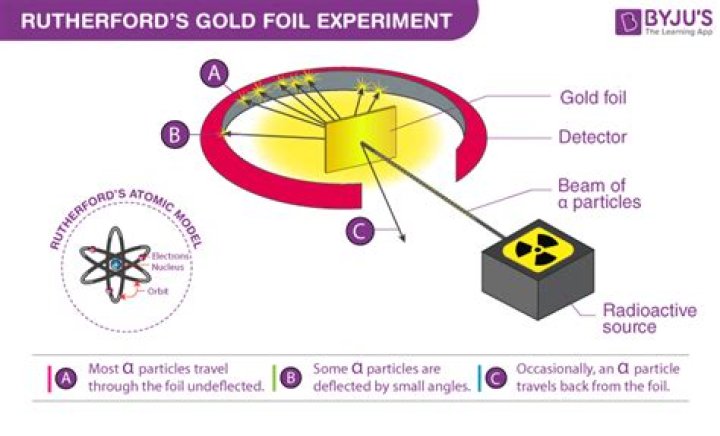
The nucleus was postulated as small and dense to account for the scattering of alpha particles from thin gold foil, as observed in a series of experiments performed by undergraduate Ernest Marsden under the direction of Rutherford and German physicist Hans Geiger in 1909.
Who discovered the number of protons?
E. Rutherford
Discovery of Electrons, Protons and Neutrons
| Discoverer | Year of Discovery | |
|---|---|---|
| Proton | E. Rutherford | 1909 |
| Neutron | James Chadwick | 1932 |
| Electron | J.J. Thomson | 1897 |
Who discovered gold number?
The Gold Number is the minimum weight (in milligrams) of a protective colloid required to prevent the coagulation of 10 ml of a standard hydro gold sol when 1 ml of a 10% sodium chloride solution is added to it. It was first used by Richard Adolf Zsigmondy.
How many protons make up gold?
79 protons
Gold is the chemical element with 79 protons in each atomic nucleus. Every atom containing 79 protons is a gold atom, and all gold atoms behave the same chemically. In principle, we can therefore create gold by simply assembling 79 protons (and enough neutrons to make the nucleus stable).
Who found the electron?
physicist J.J. Thomson
During the 1880s and ’90s scientists searched cathode rays for the carrier of the electrical properties in matter. Their work culminated in the discovery by English physicist J.J. Thomson of the electron in 1897.
What did gold foil experiment prove?
The Geiger–Marsden experiments (also called the Rutherford gold foil experiment) were a landmark series of experiments by which scientists learned that every atom has a nucleus where all of its positive charge and most of its mass is concentrated.
Who is the father of proton?
Ernest Rutherford
The proton was discovered by Ernest Rutherford in the early 1900’s. During this period, his research resulted in a nuclear reaction which led to the first ‘splitting’ of the atom, where he discovered protons. He named his discovery “protons” based on the Greek word “protos” which means first.
Who found the neutron?
James Chadwick
By 1920, physicists knew that most of the mass of the atom was located in a nucleus at its center, and that this central core contained protons. In May 1932 James Chadwick announced that the core also contained a new uncharged particle, which he called the neutron.
How many protons are in the nucleus of one atom of gold?
The number of neutrons in the nucleus of the Gold atom is the mass number of the element (the bottom numerical value) minus the atomic number, so thus the number of neutrons is 118 (196.97 was rounded off to 197). So to summarize: Number of protons is: 79.
When was the discovery of the Proton made?
Discovery of Protons The presence of positively charged particle in an atom had been first observed in 1886 by E. Goldstein based on the concept that atoms are electrically neutral i.e., it has same number of positive and negative charge.
Who was the first person to discover electrons and protons?
J.J. Thomson, an English discovered electrons in the year 1897, Eugen Goldstein, a German discovered the presence of positive particles in the year 1898, Ernest Rutherford, a New-Zealand born British discovered nucleus in 1911 and protons in 1917, and James Chadwick, an English discovered neutrons in the year 1932.
Which is the chemical symbol for the element gold?
Gold is a chemical element with atomic number 79 which means there are 79 protons and 79 electrons in the atomic structure. The chemical symbol for Gold is Au. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. The nucleus is composed of protons and neutrons.