Who discovered the positively charged nucleus using the gold foil experiment?
physicist Ernest Rutherford
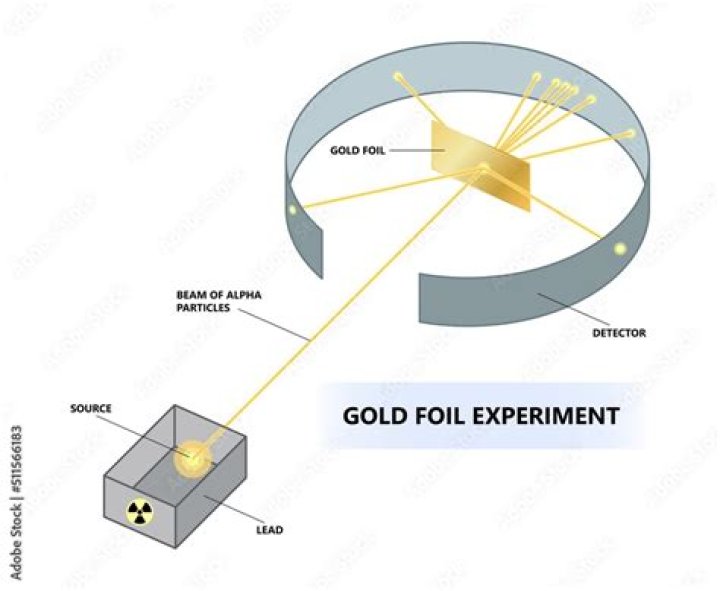
The experiments of the British physicist Ernest Rutherford in the early 20th century on the scattering of alpha particles from a thin gold foil established the Rutherford atomic model of an atom as consisting of a central, positively charged nucleus containing nearly all the mass and surrounded by a cloud of negatively …
Who concluded the gold foil experiment?
Physicist Ernest Rutherford
Physicist Ernest Rutherford established the nuclear theory of the atom with his gold-foil experiment. When he shot a beam of alpha particles at a sheet of gold foil, a few of the particles were deflected. He concluded that a tiny, dense nucleus was causing the deflections.
How did Rutherford prove the existence of the nucleus with the gold foil experiment?
Rutherford deduced that the atomic nucleus was positively charged because the alpha particles that he fired at the metal foils were positively charged, and like charges repel. In Rutherford’s experiments most of the alpha particles passed straight through the foil without being deflected.
How did the gold foil experiment prove the existence of a nucleus?
How did the gold foil experiment prove the existence of a nucleus? Rutherford’s Gold Foil Experiment proved the existance of a small massive center to atoms, which would later be known as the nucleus of an atom.
Who was the first scientist to discover the nucleus?
The atomic nucleus is the small, dense region consisting of protons and neutrons at the center of an atom, discovered in 1911 by Ernest Rutherford based on the 1909 Geiger–Marsden gold foil experiment.
When did Ernest Rutherford discover the nucleus of the atom?
Ernest Rutherford discovered the nucleus in 1911. This discovery was based on Geiger Marsden experiment or commonly known as the gold foil experiment.
How did Geiger Marsden discover the nucleus of an atom?
This discovery was based on Geiger Marsden experiment or commonly known as the gold foil experiment. He discovered that every atom contains a nucleus where its positive charge and most of its mass are concentrated. They deduced this by measuring how an alpha particle beam is scattered when it strikes a thin metal foil.