Who first did the gold foil experiment?
Rutherford
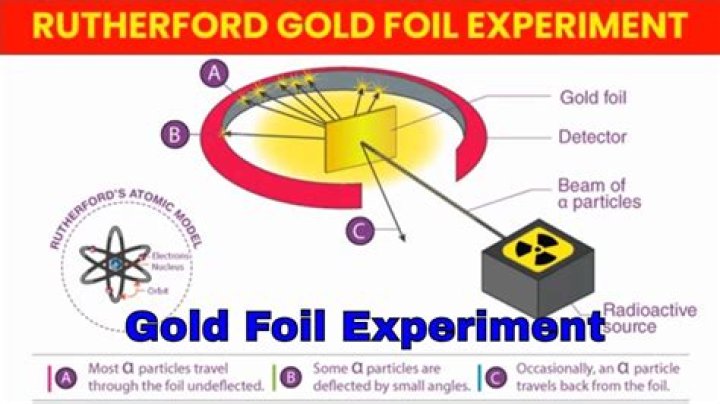
The Gold Foil Experiment. In 1911, Rutherford and coworkers Hans Geiger and Ernest Marsden initiated a series of groundbreaking experiments that would completely change the accepted model of the atom. They bombarded very thin sheets of gold foil with fast moving alpha particles.
Who did the gold foil experiment quizlet?
Rutherfords gold foil experiment involved sending positively charged alpha particles through a thin sheet of gold foil and defecting if there was any deflection of the particles. He found that most passed straight through yet some particles showed some deflection.
What was a major finding from the gold foil experiment?
Rutherford’s gold foil experiment showed that the atom is mostly empty space with a tiny, dense, positively-charged nucleus. Based on these results, Rutherford proposed the nuclear model of the atom.
What did Rutherford conclude from the gold foil experiment quizlet?
In the gold foil experiment, since some of the positive alpha particles were substantially deflected, what did Rutherford conclude? He concluded that there must be something inside an atom for the alpha particles to bounce off, that must be small, dense, and positively charged.
What was the primary outcome of Rutherford’s gold foil experiment?
The Rutherford gold leaf experiment concluded that most (99%) of all the mass of an atom is in the nucleus of the atom, that the nucleus is very small (105 times small than the size of the atom) and that is is positively charged.
What were the two main conclusions of the gold-foil experiment?
From the location and number of α-particles reaching the screen, Rutherford concluded the following: i) Almost 99% of the α-particles pass through the gold foil without any deflection. So atom must be having a lot of empty space in it. ii) Several α-particles get deflected at angles.
Who was responsible for the gold foil experiment?
Rutherford, chair of the Manchester physics department at the time of the experiment, is given primary credit for the experiment, as the theories that resulted are primarily his work. Rutherford’s gold foil experiment is also sometimes referred to as the Geiger-Marsden experiment.
What kind of foil was used in Rutherford’s experiment?
In this experiment, Rutherford used Gold Foil which was extremely thin sheet, not more than 1000 atoms thick. Gold Foil was bombarded with positively charged α-particles.
Why was gold used in the Rutherford experiment?
They used gold since it is highly malleable, producing sheets that can be only a few atoms thick, thereby ensuring smooth passage of the alpha particles. A circular screen coated with zinc sulfide surrounded the foil.
How did Rutherford’s gold foil experiment disprove the plum pudding model?
Key Questions. How did Rutherford’s gold foil experiment disprove the plum pudding model? Answer: Rutherford’s experiment showed that the atom does not contain a uniform distribution of charge. Explanation: Thomson’s plum pudding model viewed the atom as a massive blob of positive charge dotted with negative charges.